Incorporating Stakeholder Feedback in Guidelines Development for the Management of Abnormal Cervical Cancer Screening Tests
- PMID: 32243312
- PMCID: PMC7147423
- DOI: 10.1097/LGT.0000000000000524
Incorporating Stakeholder Feedback in Guidelines Development for the Management of Abnormal Cervical Cancer Screening Tests
Abstract
Objective: The 2019 ASCCP Risk-Based Management Consensus Guidelines present a paradigm shift from results- to risk-based management. Patient and provider factors can affect guideline adoption. We sought feedback from stakeholders to inform guideline development.
Materials and methods: To solicit provider feedback, we surveyed attendees at the 2019 ASCCP annual meeting regarding readiness to adopt proposed changes and used a web-based public comment period to gauge agreement/disagreement with preliminary guidelines. We elicited patient feedback via a brief survey on preferences around proposed recommendations for treatment without biopsy. Surveys and public comment included both closed-ended and free-text items. Quantitative results were analyzed using descriptive statistics; qualitative results were analyzed using content analysis. Results were incorporated into guideline development in real time.
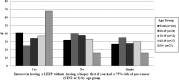
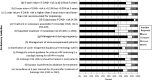
Results: Surveys indicated that 98% of providers currently evaluate their patients' past results to determine management; 88% felt formally incorporating history into management would represent an improvement in care. Most providers supported expedited treatment without biopsy: 22% currently perform expedited treatment and 60% were willing to do so. Among patients, 41% preferred expedited treatment, 32% preferred biopsy before treatment, and the remainder were undecided. Responses from the public comment period included agreement/disagreement with preliminary guidelines, reasons for disagreement, and suggestions for improvement.
Conclusions: Stakeholder feedback was incorporated into the development of the 2019 ASCCP Risk-Based Management Consensus Guidelines. Proposed recommendations with less than two-thirds agreement in the public comment period were considered for revision. Findings underscore the importance of stakeholder feedback in developing guidelines that meet the needs of patients and providers.
Conflict of interest statement
R.B.P. is a Co-Chair of the ASCCP Risk-Based Management Consensus Guidelines process. R.N., M.S., and J.L. are Co-Chairs for the Communication Working Group for the 2019 ASCCP Risk-Based Consensus Management Guidelines. T.F. is the founder of Cervivor, and served on the Treatment and Communications Working Groups for the 2019 ASCCP Risk-Based Consensus Management Guidelines. L.N.F., S.T.V., M.M., and P.L. have declared they have no conflicts of interest. The conclusions, findings, and opinions expressed by authors contributing to this journal do not necessarily reflect the official position of the US Department of Health and Human Services, the National Cancer Institute, and the Centers for Disease Control and Prevention.
Figures
Similar articles
-
A cross-sectional survey examining clinician characteristics, practices, and attitudes associated with adoption of the 2019 American Society for Colposcopy and Cervical Pathology risk-based management consensus guidelines.Cancer. 2023 Sep 1;129(17):2671-2684. doi: 10.1002/cncr.34838. Epub 2023 May 23. Cancer. 2023. PMID: 37221653
-
Moving forward-the 2019 ASCCP Risk-Based Management Consensus Guidelines for Abnormal Cervical Cancer Screening Tests and Cancer Precursors and beyond: implications and suggestions for laboratories.J Am Soc Cytopathol. 2020 Jul-Aug;9(4):291-303. doi: 10.1016/j.jasc.2020.05.002. Epub 2020 May 23. J Am Soc Cytopathol. 2020. PMID: 32565297
-
PARENTS 2 study: consensus report for parental engagement in the perinatal mortality review process.Ultrasound Obstet Gynecol. 2019 Aug;54(2):215-224. doi: 10.1002/uog.20139. Ultrasound Obstet Gynecol. 2019. PMID: 30294945 Free PMC article.
-
Clinicians' attitude towards changes in Australian National Cervical Screening Program.J Clin Virol. 2016 Mar;76 Suppl 1:S81-S87. doi: 10.1016/j.jcv.2015.11.018. Epub 2015 Nov 19. J Clin Virol. 2016. PMID: 26651602 Review.
-
Guidelines for screening and treatment of cervical disease in the adolescent.J Pediatr Adolesc Gynecol. 2004 Oct;17(5):303-11. doi: 10.1016/j.jpag.2004.07.008. J Pediatr Adolesc Gynecol. 2004. PMID: 15581775 Review.
Cited by
-
Examining the association of clinician characteristics with perceived changes in cervical cancer screening and colposcopy practice during the COVID-19 pandemic: a mixed methods assessment.Elife. 2023 Sep 1;12:e85682. doi: 10.7554/eLife.85682. Elife. 2023. PMID: 37656169 Free PMC article.
-
Confusion and anxiety in between abnormal cervical cancer screening results and colposcopy: "The land of the unknown".Patient Educ Couns. 2023 Sep;114:107810. doi: 10.1016/j.pec.2023.107810. Epub 2023 May 23. Patient Educ Couns. 2023. PMID: 37244133 Free PMC article.
-
Assessing Physician Adherence to Guidelines for Cervical Cancer Screening and Management of Abnormal Screening Results.J Low Genit Tract Dis. 2020 Oct;24(4):337-342. doi: 10.1097/LGT.0000000000000558. J Low Genit Tract Dis. 2020. PMID: 32976292 Free PMC article.
-
Factors associated with willingness to perform expedited excisional treatment for patients at high risk for cervical precancer.Gynecol Oncol Rep. 2024 Dec 9;57:101545. doi: 10.1016/j.gore.2024.101545. eCollection 2025 Feb. Gynecol Oncol Rep. 2024. PMID: 39811829 Free PMC article.
-
An Introduction to the 2019 ASCCP Risk-Based Management Consensus Guidelines.J Low Genit Tract Dis. 2020 Apr;24(2):87-89. doi: 10.1097/LGT.0000000000000531. J Low Genit Tract Dis. 2020. PMID: 32243305 Free PMC article. No abstract available.
References
-
- US Preventive Services Task Force, Curry SJ, Krist AH, Owens DK, et al. Screening for cervical cancer: US Preventive Services Task Force recommendation statement. JAMA 2018;320:674–86. - PubMed
-
- Wright TC, Jr., Cox JT, Massad LS, et al. , ASCCP-Sponsored Consensus Conference. 2001 consensus guidelines for the management of women with cervical cytological abnormalities. JAMA 2002;287:2120–9. - PubMed
-
- Wright TC, Jr., Massad LS, Dunton CJ, et al. , 2006 ASCCP-Sponsored Consensus Conference. 2006 consensus guidelines for the management of women with abnormal cervical screening tests. J Low Genit Tract Dis 2007;11:201–22. - PubMed
-
- Massad LS, Einstein MH, Huh WK, et al. , 2012 ASCCP Consensus Guidelines Conference. 2012 updated consensus guidelines for the management of abnormal cervical cancer screening tests and cancer precursors. Obstet Gynecol 2013;121:829–46. - PubMed

