Ocular cytomegalovirus latency exacerbates the development of choroidal neovascularization
- PMID: 32243583
- PMCID: PMC7356643
- DOI: 10.1002/path.5447
Ocular cytomegalovirus latency exacerbates the development of choroidal neovascularization
Abstract
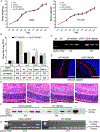
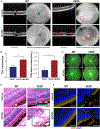
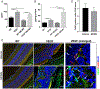
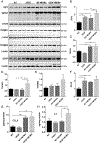
Age-related macular degeneration (AMD) is a complex, multifactorial, progressive disease which represents a leading cause of irreversible visual impairment and blindness in older individuals. Human cytomegalovirus (HCMV), which infects 50-80% of humans, is usually acquired during early life and persists in a latent state for the life of the individual. In view of its previously described pro-angiogenic properties, we hypothesized that cytomegalovirus might be a novel risk factor for progression to an advanced form, neovascular AMD, which is characterized by choroidal neovascularization (CNV). The purpose of this study was to investigate if latent ocular murine cytomegalovirus (MCMV) infection exacerbated the development of CNV in vascular endothelial growth factor (VEGF)-overexpressing VEGF-Ahyper mice. Here we show that neonatal infection with MCMV resulted in dissemination of virus to various organs throughout the body including the eye, where it localized principally to the choroid in both VEGF-overexpressingVEGF-Ahyper and wild-type(WT) 129 mice. By 6 months post-infection, no replicating virus was detected in eyes and extraocular tissues, although virus DNA was still present in all eyes and extraocular tissues of both VEGF-Ahyper and WT mice. Expression of MCMV immediate early (IE) 1 mRNA was detected only in latently infected eyes of VEGF-Ahyper mice, but not in eyes of WT mice. Significantly increased CNV was observed in eyes of MCMV-infected VEGF-Ahyper mice compared to eyes of uninfected VEGF-Ahyper mice, while no CNV lesions were observed in eyes of either infected or uninfected WT mice. Protein levels of several inflammatory/angiogenic factors, particularly VEGF and IL-6, were significantly higher in eyes of MCMV-infected VEGF-Ahyper mice, compared to uninfected controls. Initial studies of ocular tissue from human cadavers revealed that HCMV DNA was present in four choroid/retinal pigment epithelium samples from 24 cadavers. Taken together, our data suggest that ocular HCMV latency could be a significant risk factor for the development of AMD. © 2020 Pathological Society of Great Britain and Ireland. Published by John Wiley & Sons, Ltd.
Keywords: VEGF; age-related macular degeneration; choroidal neovascularization; cytomegalovirus; inflammation.
© 2020 Pathological Society of Great Britain and Ireland. Published by John Wiley & Sons, Ltd.
Conflict of interest statement
No conflicts of interest were declared.
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

