Enantioselective Pallada-Electrocatalyzed C-H Activation by Transient Directing Groups: Expedient Access to Helicenes
- PMID: 32243685
- PMCID: PMC7497116
- DOI: 10.1002/anie.202003826
Enantioselective Pallada-Electrocatalyzed C-H Activation by Transient Directing Groups: Expedient Access to Helicenes
Abstract
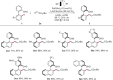
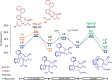
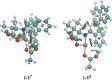
Asymmetric pallada-electrocatalyzed C-H olefinations were achieved through the synergistic cooperation with transient directing groups. The electrochemical, atroposelective C-H activations were realized with high position-, diastereo-, and enantio-control under mild reaction conditions to obtain highly enantiomerically-enriched biaryls and fluorinated N-C axially chiral scaffolds. Our strategy provided expedient access to, among others, novel chiral BINOLs, dicarboxylic acids and helicenes of value to asymmetric catalysis. Mechanistic studies by experiments and computation provided key insights into the catalyst's mode of action.
Keywords: asymmetric C−H activation; biaryls; helicene; palladium; transient directing group.
© 2020 The Authors. Published by Wiley-VCH Verlag GmbH & Co. KGaA.
Conflict of interest statement
The authors declare no conflict of interest.
Figures





References
Grants and funding
LinkOut - more resources
Full Text Sources

