Programmed Cell Death-1: Programmed Cell Death-Ligand 1 Interaction Protects Human Cardiomyocytes Against T-Cell Mediated Inflammation and Apoptosis Response In Vitro
- PMID: 32244307
- PMCID: PMC7177768
- DOI: 10.3390/ijms21072399
Programmed Cell Death-1: Programmed Cell Death-Ligand 1 Interaction Protects Human Cardiomyocytes Against T-Cell Mediated Inflammation and Apoptosis Response In Vitro
Abstract
Aim: Immunological checkpoint therapy is considered a powerful method for cancer therapy and acts by re-activating autologous T cells to kill the cancer cell. Myocarditis cases have been reported in cancer patients after immunological therapy; for example, nivolumab treatment is a monoclonal antibody that blocks programmed cell death-1/programmed cell death ligand-1 ligand interaction. This project provided insight into the inflammatory response as a benchmark to investigate the potential cardiotoxic effect of T cell response to the programmed cell death-1 (PD-1)/programmed cell death ligand-1 (PD-L1) axis in regulating cardiomyocyte injury in vitro.
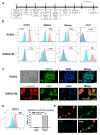
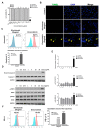
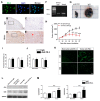
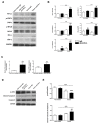
Methods and results: We investigated cardiomyopathy resulted from the PD-1/PD-L1 axis blockade using the anti-PD-1 antibody in Rockefeller University embryonic stem cells-derived cardiomyocytes (RUES2-CMs) and a melanoma tumor-bearing murine model. We found that nivolumab alone did not induce inflammatory-related proteins, including PD-L1 expression, and did not induce apoptosis, which was contrary to doxorubicin, a cardiotoxic chemotherapy drug. However, nivolumab was able to exacerbate the immune response by increasing cytokine and inflammatory gene expression in RUES2-CMs when co-cultured with CD4+ T lymphocytes and induced apoptosis. This effect was not observed when RUES2-CMs were co-cultured with CD8+ T lymphocytes. The in vivo model showed that the heart function of tumor-bearing mice was decreased after treatment with anti-PD-1 antibody and demonstrated a dilated left ventricle histological examination. The dilated left ventricle was associated with an infiltration of CD4+ and CD8+ T lymphocytes into the myocardium. PD-L1 and inflammatory-associated gene expression were significantly increased in anti-PD-1-treated tumor-bearing mice. Cleaved caspase-3 and mouse plasma cardiac troponin I expressions were increased significantly.
Conclusion: PD-L1 expression on cardiomyocytes suppressed T-cell function. Blockade of PD-1 by nivolumab enhanced cardiomyocyte inflammation and apoptosis through the enhancement of T-cell response towards cardiomyocytes.
Keywords: PD-1; PD-L1; T-lymphocytes; human embryonic stem cell-derived cardiomyocytes; immune-related cardiotoxicity; nivolumab.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

