Antiviral, Immunomodulatory and Antiproliferative Activities of Recombinant Soluble IFNAR2 without IFN-ß Mediation
- PMID: 32244308
- PMCID: PMC7230527
- DOI: 10.3390/jcm9040959
Antiviral, Immunomodulatory and Antiproliferative Activities of Recombinant Soluble IFNAR2 without IFN-ß Mediation
Abstract
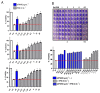
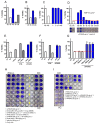
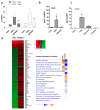
Soluble receptors of cytokines are able to modify cytokine activities and therefore the immune system, and some have intrinsic biological activities without mediation from their cytokines. The soluble interferon beta (IFN-ß) receptor is generated through alternative splicing of IFNAR2 and has both agonist and antagonist properties for IFN-ß, but its role is unknown. We previously demonstrated that a recombinant human soluble IFN-ß receptor showed intrinsic therapeutic efficacy in a mouse model of multiple sclerosis. Here we evaluate the potential biological activities of recombinant sIFNAR2 without the mediation of IFN-ß in human cells. Recombinant sIFNAR2 down-regulated the production of IL-17 and IFN-ɣ and reduced the cell proliferation rate. Moreover, it showed a strong antiviral activity, fully protecting the cell monolayer after being infected by the virus. Specific inhibitors completely abrogated the antiviral activity of IFN-ß, but not that of the recombinant sIFNAR2, and there was no activation of the JAK-STAT signaling pathway. Consequently, r-sIFNAR2 exerts immunomodulatory, antiproliferative and antiviral activities without IFN-ß mediation, and could be a promising treatment against viral infections and immune-mediated diseases.
Keywords: IFNAR; immunology; interferon; soluble receptors.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

