Investigating Algal Communities in Lacustrine and Hydro-Terrestrial Environments of East Antarctica Using Deep Amplicon Sequencing
- PMID: 32244517
- PMCID: PMC7232531
- DOI: 10.3390/microorganisms8040497
Investigating Algal Communities in Lacustrine and Hydro-Terrestrial Environments of East Antarctica Using Deep Amplicon Sequencing
Abstract
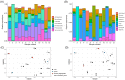
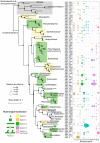
Antarctica has one of the most extreme environments on Earth, with low temperatures and low nutrient levels. Antarctica's organisms live primarily in the coastal, ice-free areas which cover approximately 0.18% of the continent's surface. Members of Cyanobacteria and eukaryotic algae are important primary producers in Antarctica since they can synthesize organic compounds from carbon dioxide and water using solar energy. However, community structures of photosynthetic algae in Antarctica have not yet been fully explored at molecular level. In this study, we collected diverse algal samples in lacustrine and hydro-terrestrial environments of Langhovde and Skarvsnes, which are two ice-free regions in East Antarctica. We performed deep amplicon sequencing of both 16S ribosomal ribonucleic acid (rRNA) and 18S rRNA genes, and we explored the distribution of sequence variants (SVs) of these genes at single nucleotide difference resolution. SVs of filamentous Cyanobacteria genera, including Leptolyngbya, Pseudanabaena, Phormidium, Nodosilinea, Geitlerinama, and Tychonema, were identified in most of the samples, whereas Phormidesmis SVs were distributed in fewer samples. We also detected unicellular, multicellular or heterocyst forming Cyanobacteria strains, but in relatively small abundance. For SVs of eukaryotic algae, Chlorophyta, Cryptophyta, and Ochrophyta were widely distributed among the collected samples. In addition, there was a red colored bloom of eukaryotic alga, Geminigera cryophile (Cryptophyta), in the Langhovde coastal area. Eukaryotic SVs of Acutuncus antarcticus and/or Diphascon pingue of Tardigrada were dominant among most of the samples. Our data revealed the detailed structures of the algal communities in Langhovde and Skarvsnes. This will contribute to our understanding of Antarctic ecosystems and support further research into this subject.
Keywords: Algae; Antarctica; Cyanobacteria; Hydro-Terrestrial; Lacustrine; Microbiome; Tardigrade.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures



Similar articles
-
Microbial community composition of terrestrial habitats in East Antarctica with a focus on microphototrophs.Front Microbiol. 2024 Jan 5;14:1323148. doi: 10.3389/fmicb.2023.1323148. eCollection 2023. Front Microbiol. 2024. PMID: 38249463 Free PMC article.
-
Bacterial and eukaryotic biodiversity patterns in terrestrial and aquatic habitats in the Sør Rondane Mountains, Dronning Maud Land, East Antarctica.FEMS Microbiol Ecol. 2016 Jun;92(6):fiw041. doi: 10.1093/femsec/fiw041. Epub 2016 Mar 2. FEMS Microbiol Ecol. 2016. PMID: 26936447
-
On the use of high-throughput sequencing for the study of cyanobacterial diversity in Antarctic aquatic mats.J Phycol. 2016 Jun;52(3):356-68. doi: 10.1111/jpy.12399. Epub 2016 Apr 27. J Phycol. 2016. PMID: 27273529
-
Trace metals in Antarctica related to climate change and increasing human impact.Rev Environ Contam Toxicol. 2000;166:129-73. Rev Environ Contam Toxicol. 2000. PMID: 10868078 Review.
-
Phytoplankton communities of polar regions--Diversity depending on environmental conditions and chemical anthropopressure.J Environ Manage. 2016 Apr 15;171:243-259. doi: 10.1016/j.jenvman.2016.01.026. Epub 2016 Feb 2. J Environ Manage. 2016. PMID: 26846983 Review.
Cited by
-
Genome sequencing of the NIES Cyanobacteria collection with a focus on the heterocyst-forming clade.DNA Res. 2021 Oct 11;28(6):dsab024. doi: 10.1093/dnares/dsab024. DNA Res. 2021. PMID: 34677568 Free PMC article.
-
A specific combination of dual index adaptors decreases the sensitivity of amplicon sequencing with the Illumina platform.DNA Res. 2020 Aug 1;27(4):dsaa017. doi: 10.1093/dnares/dsaa017. DNA Res. 2020. PMID: 32810209 Free PMC article.
-
Distinct prokaryotic and eukaryotic communities and networks in two agricultural fields of central Japan with different histories of maize-cabbage rotation.Sci Rep. 2023 Sep 18;13(1):15435. doi: 10.1038/s41598-023-42291-y. Sci Rep. 2023. PMID: 37723228 Free PMC article.
-
DNA Metabarcoding Focusing on Eukaryote Communities on Langhovde Glacier, East Antarctica.Environ Microbiol Rep. 2025 Aug;17(4):e70117. doi: 10.1111/1758-2229.70117. Environ Microbiol Rep. 2025. PMID: 40602625 Free PMC article.
References
-
- Burton-Johnson A., Black M., Fretwell P.T., Kaluza-Gilbert J. An automated methodology for differentiating rock from snow, clouds and sea in Antarctica from Landsat 8 imagery: A new rock outcrop map and area estimation for the entire Antarctic continent. Cryosphere. 2016;10:1665–1677. doi: 10.5194/tc-10-1665-2016. - DOI
-
- Vincent W.F., Quesada A. Cyanobacteria in high latitude lakes, rivers and seas. In: Whitton B.A., editor. Ecology of Cyanobacteria II. Springer; Dordrecht, The Netherlands: 2012. pp. 371–385.
-
- Broady P.A. Diversity, distribution and dispersal of Antarctic terrestrial algae. Biodivers. Conserv. 1996;5:1307–1335. doi: 10.1007/BF00051981. - DOI
-
- Seckbach J. Algae and Cyanobacteria in Extreme Environments. Springer; Dordrecht, The Netherlands: 2007. p. 813.
Grants and funding
LinkOut - more resources
Full Text Sources

