Characterization of Carbonic Anhydrase In Vivo Using Magnetic Resonance Spectroscopy
- PMID: 32244610
- PMCID: PMC7178054
- DOI: 10.3390/ijms21072442
Characterization of Carbonic Anhydrase In Vivo Using Magnetic Resonance Spectroscopy
Abstract
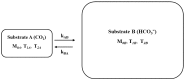
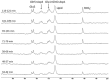
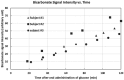
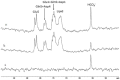
Carbonic anhydrase is a ubiquitous metalloenzyme that catalyzes the reversible interconversion of CO2/HCO3-. Equilibrium of these species is maintained by the action of carbonic anhydrase. Recent advances in magnetic resonance spectroscopy have allowed, for the first time, in vivo characterization of carbonic anhydrase in the human brain. In this article, we review the theories and techniques of in vivo 13C magnetization (saturation) transfer magnetic resonance spectroscopy as they are applied to measuring the rate of exchange between CO2 and HCO3- catalyzed by carbonic anhydrase. Inhibitors of carbonic anhydrase have a wide range of therapeutic applications. Role of carbonic anhydrases and their inhibitors in many diseases are also reviewed to illustrate future applications of in vivo carbonic anhydrase assessment by magnetic resonance spectroscopy.
Keywords: GABAergic transmission; carbonic anhydrase; in vivo MRS; neurological diseases; psychiatric diseases.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
13C saturation transfer effect of carbon dioxide-bicarbonate exchange catalyzed by carbonic anhydrase in vivo.Magn Reson Med. 2008 Mar;59(3):492-8. doi: 10.1002/mrm.21501. Magn Reson Med. 2008. PMID: 18224701
-
Investigation of the system copper(II) carbonic anhydrase and HCO3-/CO2.J Inorg Biochem. 1983 Jun;18(3):221-9. doi: 10.1016/0162-0134(83)85004-1. J Inorg Biochem. 1983. PMID: 6409996
-
A 13C nuclear magnetic resonance study of CO2/HCO-3 exchange catalyzed by human carbonic anhydrase I.Eur J Biochem. 1982 Dec;129(1):165-9. doi: 10.1111/j.1432-1033.1982.tb07035.x. Eur J Biochem. 1982. PMID: 6819139
-
The many types of carbonic anhydrases in photosynthetic organisms.Plant Sci. 2018 Mar;268:11-17. doi: 10.1016/j.plantsci.2017.12.002. Epub 2017 Dec 7. Plant Sci. 2018. PMID: 29362079 Review.
-
Role of Carbonic Anhydrase in Cerebral Ischemia and Carbonic Anhydrase Inhibitors as Putative Protective Agents.Int J Mol Sci. 2021 May 10;22(9):5029. doi: 10.3390/ijms22095029. Int J Mol Sci. 2021. PMID: 34068564 Free PMC article. Review.
Cited by
-
Role of the Hypoxic-Secretome in Seed and Soil Metastatic Preparation.Cancers (Basel). 2022 Nov 30;14(23):5930. doi: 10.3390/cancers14235930. Cancers (Basel). 2022. PMID: 36497411 Free PMC article. Review.
-
Molecular Mechanisms and Clinical Aspects of Colitis-Associated Cancer in Ulcerative Colitis.Cells. 2025 Jan 22;14(3):162. doi: 10.3390/cells14030162. Cells. 2025. PMID: 39936954 Free PMC article. Review.
-
Carbonic anhydrase activity in the frontal lobe of human brain.NMR Biomed. 2021 Jun;34(6):e4501. doi: 10.1002/nbm.4501. Epub 2021 Mar 8. NMR Biomed. 2021. PMID: 33682938 Free PMC article.
-
Carbonic Anhydrase and Biomarker Research: New Insights.Int J Mol Sci. 2023 Jun 2;24(11):9687. doi: 10.3390/ijms24119687. Int J Mol Sci. 2023. PMID: 37298637 Free PMC article.
References
-
- Aspatwar A., Tolvanen M.E.E., Ortutay C., Parkkila S. Carbonic anhydrase related proteins: Molecular biology and evolution. Subcell Biochem. 2014;75:135–156. - PubMed
-
- Tashian R.E., Hewett-Emmett D., Carter N., Bergenhem N.C.H. Carbonic anhydrase (CA)-related proteins (CA-RPs) and transmembrane proteins with CA or CA-RP domains. In: Chegwidden W.R., Carter N.D., Edwards Y.H., editors. The carbonic Anhydrases: New Horizons. Birkhäuser; Basel, Switzerland: 2000. pp. 105–120. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

