Potential Adverse Public Health Effects Afforded by the Ingestion of Dietary Lipid Oxidation Product Toxins: Significance of Fried Food Sources
- PMID: 32244669
- PMCID: PMC7254282
- DOI: 10.3390/nu12040974
Potential Adverse Public Health Effects Afforded by the Ingestion of Dietary Lipid Oxidation Product Toxins: Significance of Fried Food Sources
Abstract
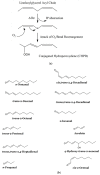
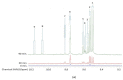
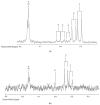
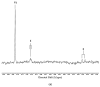
Exposure of polyunsaturated fatty acid (PUFA)-rich culinary oils (COs) to high temperature frying practices generates high concentrations of cytotoxic and genotoxic lipid oxidation products (LOPs) via oxygen-fueled, recycling peroxidative bursts. These toxins, including aldehydes and epoxy-fatty acids, readily penetrate into fried foods and hence are available for human consumption; therefore, they may pose substantial health hazards. Although previous reports have claimed health benefits offered by the use of PUFA-laden COs for frying purposes, these may be erroneous in view of their failure to consider the negating adverse public health threats presented by food-transferable LOPs therein. When absorbed from the gastrointestinal (GI) system into the systemic circulation, such LOPs may significantly contribute to enhanced risks of chronic non-communicable diseases (NCDs), e.g. cancer, along with cardiovascular and neurological diseases. Herein, we provide a comprehensive rationale relating to the public health threats posed by the dietary ingestion of LOPs in fried foods. We begin with an introduction to sequential lipid peroxidation processes, describing the noxious effects of LOP toxins generated therefrom. We continue to discuss GI system interactions, the metabolism and biotransformation of primary lipid hydroperoxide LOPs and their secondary products, and the toxicological properties of these agents, prior to providing a narrative on chemically-reactive, secondary aldehydic LOPs available for human ingestion. In view of a range of previous studies focused on their deleterious health effects in animal and cellular model systems, some emphasis is placed on the physiological fate of the more prevalent and toxic α,β-unsaturated aldehydes. We conclude with a description of targeted nutritional and interventional strategies, whilst highlighting the urgent and unmet clinical need for nutritional and epidemiological trials probing relationships between the incidence of NCDs, and the frequency and estimated quantities of dietary LOP intake.
Keywords: acrolein; aldehyde toxins; atherosclerosis; cancer; cooking oil fumes; cytogenicity/gentoxicity/mutagenicity; fried foods; frying oils; lipid hydroperoxides; lipid oxidation products; maximum human dietary intake (MHDI).
Conflict of interest statement
None of the authors declare any conflicts of interest.
Figures





Similar articles
-
Evidence-Based Challenges to the Continued Recommendation and Use of Peroxidatively-Susceptible Polyunsaturated Fatty Acid-Rich Culinary Oils for High-Temperature Frying Practises: Experimental Revelations Focused on Toxic Aldehydic Lipid Oxidation Products.Front Nutr. 2022 Jan 5;8:711640. doi: 10.3389/fnut.2021.711640. eCollection 2021. Front Nutr. 2022. PMID: 35071288 Free PMC article. Review.
-
Toxic aldehyde generation in and food uptake from culinary oils during frying practices: peroxidative resistance of a monounsaturate-rich algae oil.Sci Rep. 2019 Mar 11;9(1):4125. doi: 10.1038/s41598-019-39767-1. Sci Rep. 2019. PMID: 30858398 Free PMC article.
-
Comparative 1H NMR-Based Chemometric Evaluations of the Time-Dependent Generation of Aldehydic Lipid Oxidation Products in Culinary Oils Exposed to Laboratory-Simulated Shallow Frying Episodes: Differential Patterns Observed for Omega-3 Fatty Acid-Containing Soybean Oils.Foods. 2021 Oct 17;10(10):2481. doi: 10.3390/foods10102481. Foods. 2021. PMID: 34681530 Free PMC article.
-
Effectiveness of different antioxidants in suppressing the evolution of thermally induced peroxidation products in hemp seed oil.Food Res Int. 2024 Jul;188:114415. doi: 10.1016/j.foodres.2024.114415. Epub 2024 May 3. Food Res Int. 2024. PMID: 38823855
-
Dietary glycation compounds - implications for human health.Crit Rev Toxicol. 2024 Sep;54(8):485-617. doi: 10.1080/10408444.2024.2362985. Epub 2024 Aug 16. Crit Rev Toxicol. 2024. PMID: 39150724
Cited by
-
1H NMR Study of the In Vitro Digestion of Highly Oxidized Soybean Oil and the Effect of the Presence of Ovalbumin.Foods. 2021 Jul 6;10(7):1573. doi: 10.3390/foods10071573. Foods. 2021. PMID: 34359443 Free PMC article.
-
Evaluation of Steady-State and Time-Resolved Fluorescence Spectroscopy as a Method for Assessing the Impact of Photo-Oxidation on Refined Soybean Oils.Foods. 2023 Apr 30;12(9):1862. doi: 10.3390/foods12091862. Foods. 2023. PMID: 37174400 Free PMC article.
-
Quality and Nutritional Changes of Traditional Cupcakes in the Processing and Storage as a Result of Sunflower Oil Replacements with Refined Olive Pomace Oil.Foods. 2023 May 24;12(11):2125. doi: 10.3390/foods12112125. Foods. 2023. PMID: 37297368 Free PMC article.
-
Vegetable Oils and Their Use for Frying: A Review of Their Compositional Differences and Degradation.Foods. 2024 Dec 23;13(24):4186. doi: 10.3390/foods13244186. Foods. 2024. PMID: 39767128 Free PMC article. Review.
-
Analysis of the Generation of Harmful Aldehydes in Edible Oils During Sunlight Exposure and Deep-Frying Using High-Field Proton Nuclear Magnetic Resonance Spectroscopy.Foods. 2025 Feb 5;14(3):513. doi: 10.3390/foods14030513. Foods. 2025. PMID: 39942106 Free PMC article.
References
-
- Grootveld M., Ruiz-Rodado V., Silwood C.J.L. Detection, monitoring and deleterious health effects of lipid oxidation products generated in COs during thermal stressing episodes. Inform Am. Oil Chem. Soc. 2014;25:614–624.
-
- Grootveld M., Silwood C.J.L., Addis P., Claxson A.W.D., Bonet Serra B., Viana M. Health effects of oxidised heated oils. Foodserv. Res. Internat. 2001;13:39–53. doi: 10.1111/j.1745-4506.2001.tb00028.x. - DOI
-
- Estévez M., Li Z., Soladoye O.P., Van-Hecke T. Chapter Two - Health Risks of Food Oxidation. Advanc. Food Nutrit. Res. 2017;82:45–81. - PubMed
-
- Jackson V., Penumetcha M. Dietary oxidised lipids, health consequences and novel food technologies that thwart food lipid oxidation: An update. Intern. J. Food Sci. Technol. 2019;54:1981–1988. doi: 10.1111/ijfs.14058. - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

