Viral Pathogenesis, Recombinant Vaccines, and Oncolytic Virotherapy: Applications of the Canine Distemper Virus Reverse Genetics System
- PMID: 32244946
- PMCID: PMC7150803
- DOI: 10.3390/v12030339
Viral Pathogenesis, Recombinant Vaccines, and Oncolytic Virotherapy: Applications of the Canine Distemper Virus Reverse Genetics System
Abstract
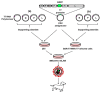
Canine distemper virus (CDV) is a highly contagious pathogen transmissible to a broad range of terrestrial and aquatic carnivores. Despite the availability of attenuated vaccines against CDV, the virus remains responsible for outbreaks of canine distemper (CD) with significant morbidity and mortality in domesticated and wild carnivores worldwide. CDV uses the signaling lymphocytic activation molecule (SLAM, or CD150) and nectin-4 (PVRL4) as entry receptors, well-known tumor-associated markers for several lymphadenomas and adenocarcinomas, which are also responsible for the lysis of tumor cells and apparent tumor regression. Thus, CDV vaccine strains have emerged as a promising platform of oncolytic viruses for use in animal cancer therapy. Recent advances have revealed that use of the CDV reverse genetic system (RGS) has helped increase the understanding of viral pathogenesis and explore the development of recombinant CDV vaccines. In addition, genetic engineering of CDV based on RGS approaches also has the potential of enhancing oncolytic activity and selectively targeting tumors. Here, we reviewed the host tropism and pathogenesis of CDV, and current development of recombinant CDV-based vaccines as well as their use as oncolytic viruses against cancers.
Keywords: canine distemper virus; pathogenesis; recombinant vaccine; reverse genetic system; virotherapy.
Conflict of interest statement
The authors declare no conflicts of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures

References
-
- Woo P.C., Lau S.K., Wong B.H., Fan R.Y., Wong A.Y., Zhang A.J., Wu Y., Choi G.K., Li K.S., Hui J., et al. Feline morbillivirus, a previously undescribed paramyxovirus associated with tubulointerstitial nephritis in domestic cats. Proc. Natl. Acad. Sci. USA. 2012;109:5435–5440. doi: 10.1073/pnas.1119972109. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

