The Dynamics of P Granule Liquid Droplets Are Regulated by the Caenorhabditis elegans Germline RNA Helicase GLH-1 via Its ATP Hydrolysis Cycle
- PMID: 32245789
- PMCID: PMC7268986
- DOI: 10.1534/genetics.120.303052
The Dynamics of P Granule Liquid Droplets Are Regulated by the Caenorhabditis elegans Germline RNA Helicase GLH-1 via Its ATP Hydrolysis Cycle
Abstract
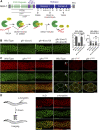
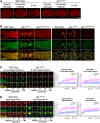
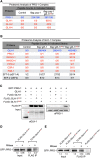
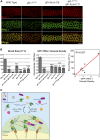
P granules are phase-separated liquid droplets that play important roles in the maintenance of germ cell fate in Caenorhabditis elegans Both the localization and formation of P granules are highly dynamic, but mechanisms that regulate such processes remain poorly understood. Here, we show evidence that the VASA-like germline RNA helicase GLH-1 couples distinct steps of its ATPase hydrolysis cycle to control the formation and disassembly of P granules. In addition, we found that the phenylalanine-glycine-glycine repeats in GLH-1 promote its localization at the perinucleus. Proteomic analyses of the GLH-1 complex with a GLH-1 mutation that interferes with P granule disassembly revealed transient interactions of GLH-1 with several Argonautes and RNA-binding proteins. Finally, we found that defects in recruiting the P granule component PRG-1 to perinuclear foci in the adult germline correlate with the fertility defects observed in various GLH-1 mutants. Together, our results highlight the versatile roles of an RNA helicase in controlling the formation of liquid droplets in space and time.
Keywords: ATP hydrolysis; Germ granule; P granule; RNA helicase; VASA; phase separation.
Copyright © 2020 by the Genetics Society of America.
Figures
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

