Superior colliculus modulates cortical coding of somatosensory information
- PMID: 32245963
- PMCID: PMC7125203
- DOI: 10.1038/s41467-020-15443-1
Superior colliculus modulates cortical coding of somatosensory information
Abstract
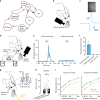
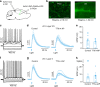
The cortex modulates activity in superior colliculus via a direct projection. What is largely unknown is whether (and if so how) the superior colliculus modulates activity in the cortex. Here, we investigate this issue and show that optogenetic activation of superior colliculus changes the input-output relationship of neurons in somatosensory cortex, enhancing responses to low amplitude whisker deflections. While there is no direct pathway from superior colliculus to somatosensory cortex, we found that activation of superior colliculus drives spiking in the posterior medial (POm) nucleus of the thalamus via a powerful monosynaptic pathway. Furthermore, POm neurons receiving input from superior colliculus provide monosynaptic excitatory input to somatosensory cortex. Silencing POm abolished the capacity of superior colliculus to modulate cortical whisker responses. Our findings indicate that the superior colliculus, which plays a key role in attention, modulates sensory processing in somatosensory cortex via a powerful di-synaptic pathway through the thalamus.
Conflict of interest statement
The authors declare no competing interests.
Figures





References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

