Anti-HIV agent azidothymidine decreases Tet(X)-mediated bacterial resistance to tigecycline in Escherichia coli
- PMID: 32246108
- PMCID: PMC7125129
- DOI: 10.1038/s42003-020-0877-5
Anti-HIV agent azidothymidine decreases Tet(X)-mediated bacterial resistance to tigecycline in Escherichia coli
Abstract
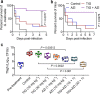
Recent emergence of high-level tigecycline resistance mediated by Tet(X3/X4) in Enterobacteriaceae undoubtably constitutes a serious threat for public health worldwide. Antibiotic adjuvant strategy makes antibiotic more effective against these resistant pathogens through interfering intrinsic resistance mechanisms or enhancing antibiotic actions. Herein, we screened a collection of drugs to identify compounds that are able to restore tigecycline activity against resistant pathogens. Encouragingly, we discovered that anti-HIV agent azidothymidine dramatically potentiates tigecycline activity against clinically resistant bacteria. Meanwhile, addition of azidothymidine prevents the evolution of tigecycline resistance in E. coli and the naturally occurring horizontal transfer of tet(X4). Evidence demonstrated that azidothymidine specifically inhibits DNA synthesis and suppresses resistance enzyme activity. Moreover, in in vivo infection models by Tet(X4)-expression E. coli, the combination of azidothymidine and tigecycline achieved remarkable treatment benefits including increased survival and decreased bacterial burden. These findings provide an effective regimen to treat infections caused by tigecycline-resistant Escherichia coli.
Conflict of interest statement
The authors declare no competing interests.
Figures



References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

