Epicutaneous sensitization in the development of food allergy: What is the evidence and how can this be prevented?
- PMID: 32249942
- PMCID: PMC7494573
- DOI: 10.1111/all.14304
Epicutaneous sensitization in the development of food allergy: What is the evidence and how can this be prevented?
Abstract
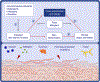
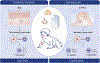
There is increasing evidence regarding the importance of allergic sensitization through the skin. In this review, we provide an overview of the atopic march and immune mechanism underlying the sensitization and effector phase of food allergy. We present experimental models and human data that support the concept of epicutaneous sensitization and how this forms one half of the dual-allergen exposure hypothesis. We discuss specific important elements in the skin (FLG and other skin barrier gene mutations, Langerhans cells, type 2 innate lymphoid cells, IL-33, TSLP) that have important roles in the development of allergic responses as well as the body of evidence on environmental allergen exposure and how this can sensitize an individual. Given the link between skin barrier impairment, atopic dermatitis, food allergy, allergic asthma, and allergic rhinitis, it is logical that restoring the skin barrier and prevention or treating atopic dermatitis would have beneficial effects on prevention of related allergic diseases, particularly food allergy. We present the experimental and human studies that have evaluated this approach and discuss various factors which may influence the success of these approaches, such as the type of emollient chosen for the intervention, the role of managing skin inflammation, and differences between primary and secondary prevention of atopic dermatitis to achieve the desired outcome.
Keywords: atopic dermatitis; cutaneous sensitization; emollient; filaggrin gene; food allergy.
© 2020 The Authors. Allergy published by European Academy of Allergy and Clinical Immunology and John Wiley & Sons Ltd.
Conflict of interest statement
Conflict of Interest Statement
Dr. Brough reports personal fees from DBV Technologies, and non-financial support from ThermoScientific; Dr. Nadeau reports grants from National Institute of Allergy and Infectious Diseases (NIAID), Food Allergy Research & Education (FARE), End Allergies Together (EAT), Allergenis, and Ukko Pharma; Grant awardee at NIAID, National Institute of Environmental Health Sciences (NIEHS), National Heart, Lung, and Blood Institute (NHLBI), and the Environmental Protection Agency (EPA); is involved in Clinical trials with Regeneron, Genentech, AImmune Therapeutics, DBV Technologies, AnaptysBio, Adare Pharmaceuticals, and Stallergenes-Greer; Research Sponsorship by Novartis, Sanofi, Astellas, Nestle; Data and Safety Monitoring Board member at Novartis and NHLBI; Cofounded Before Brands, Alladapt, ForTra, and Iggenix; Chief Intellectual Office at FARE, Director of the World Allergy Organization (WAO) Center of Excellence at Stanford, Personal fees from Regeneron, Astrazeneca, ImmuneWorks, and Cour Pharmaceuticals; Consultant and Advisory Board Member at Ukko, Before Brands, Alladapt, IgGenix, Probio, Vedanta, Centecor, Seed, Novartis, NHBLI, EPA, National Scientific Committee of ITN and NIH Programs; US patents (patent numbers 62/647,389; 62/119,014; 12/610,940, 12/686,121, 10/064,936, 62/767,444; application numbers S10–392); Dr. Sindher reports grants from Aimmune, DBV Technologies, and Regeneron; Dr. Chan reports non-financial support from Novartis and grants from Aimmune; Dr. Bahnson reports personal fees from King’s College and DBV technologies; Dr. Lack reports grants from National Institute of Allergy and Infectious
Diseases (NIAID, NIH), Food Allergy & Research Education (FARE), MRC & Asthma UK Centre, UK Dept of Health through NIHR, National Peanut Board (NPB), UK Food Standards Agency (FSA), The Davis Foundation, Scientific Advisory Board member and Stock Options at DBV Technologies, Shareholder at Mighty Mission Me, personal fees/consultancy from Novartis, personal fees/consultancy from Sanofi-Genyzme, personal fees/consultancy from Regeneron, personal fees and consultancy from ALK-Abello; Drs. Alkotob and Leung have nothing to disclose
Figures




Similar articles
-
Mechanisms for initiation of food allergy by skin pre-disposed to atopic dermatitis.Immunol Rev. 2024 Sep;326(1):151-161. doi: 10.1111/imr.13367. Epub 2024 Jul 15. Immunol Rev. 2024. PMID: 39007725 Review.
-
Epicutaneous sensitization to food allergens in atopic dermatitis: What do we know?Pediatr Allergy Immunol. 2020 Jan;31(1):7-18. doi: 10.1111/pai.13127. Epub 2019 Oct 9. Pediatr Allergy Immunol. 2020. PMID: 31541586 Review.
-
Thymic stromal lymphopoietin rather than IL-33 drives food allergy after epicutaneous sensitization to food allergen.J Allergy Clin Immunol. 2023 Jun;151(6):1660-1666.e4. doi: 10.1016/j.jaci.2023.02.025. Epub 2023 Mar 4. J Allergy Clin Immunol. 2023. PMID: 36878383 Free PMC article.
-
Prevention of allergic disease in childhood: clinical and epidemiological aspects of primary and secondary allergy prevention.Pediatr Allergy Immunol. 2004 Jun;15 Suppl 16:4-5, 9-32. doi: 10.1111/j.1399-3038.2004.0148b.x. Pediatr Allergy Immunol. 2004. PMID: 15125698 Review.
-
Skin as the target for allergy prevention and treatment.Ann Allergy Asthma Immunol. 2024 Aug;133(2):133-143. doi: 10.1016/j.anai.2023.12.030. Epub 2024 Jan 20. Ann Allergy Asthma Immunol. 2024. PMID: 38253125 Review.
Cited by
-
New Mechanistic Advances in FcεRI-Mast Cell-Mediated Allergic Signaling.Clin Rev Allergy Immunol. 2022 Dec;63(3):431-446. doi: 10.1007/s12016-022-08955-9. Epub 2022 Oct 17. Clin Rev Allergy Immunol. 2022. PMID: 36251242 Free PMC article. Review.
-
Allergy-associated biomarkers in early life identified by Omics techniques.Front Allergy. 2024 Feb 23;5:1359142. doi: 10.3389/falgy.2024.1359142. eCollection 2024. Front Allergy. 2024. PMID: 38464396 Free PMC article. Review.
-
Phenotypes of Atopic Dermatitis and Development of Allergic Diseases.JAMA Netw Open. 2025 Jun 2;8(6):e2515094. doi: 10.1001/jamanetworkopen.2025.15094. JAMA Netw Open. 2025. PMID: 40504529 Free PMC article.
-
The Sensitization Profile for Selected Food Allergens in Polish Children Assessed with the Use of a Precision Allergy Molecular Diagnostic Technique.Int J Mol Sci. 2024 Jan 9;25(2):825. doi: 10.3390/ijms25020825. Int J Mol Sci. 2024. PMID: 38255900 Free PMC article.
-
A Narrative Mini Review on Current Status of Hypoallergenic Wheat Development for IgE-Mediated Wheat Allergy, Wheat-Dependent Exercise-Induced Anaphylaxis.Foods. 2023 Feb 23;12(5):954. doi: 10.3390/foods12050954. Foods. 2023. PMID: 36900471 Free PMC article.
References
-
- Martin PE, Koplin JJ, Eckert JK, Lowe AJ, Ponsonby AL, Osborne NJ, et al. The prevalence and socio-demographic risk factors of clinical eczema in infancy: a population-based observational study. Clin Exp Allergy 2013;43:642–651. - PubMed
-
- Wollenberg A, Barbarot S, Bieber T, Christen-Zaech S, Deleuran M, Fink-Wagner A, et al. Consensus-based European guidelines for treatment of atopic eczema (atopic dermatitis) in adults and children: part I. J Eur Acad Dermatol Venereol 2018;32:657–682. - PubMed
-
- Borna E, Nwaru BI, Bjerg A, Mincheva R, Radinger M, Lundback B, et al. Changes in the prevalence of asthma and respiratory symptoms in western Sweden between 2008 and 2016. Allergy 2019;74:1703–1715. - PubMed
-
- Global Initiative for Asthma. Global strategy for asthma management and prevention (2018 update). 2018. Available from: https://ginasthma.org/wp-content/uploads/2019/06/GINA-2019-main-report-J.... Accessed Feb 24, 2020
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

