Methylxanthines Inhibit Primary Amine Oxidase and Monoamine Oxidase Activities of Human Adipose Tissue
- PMID: 32252407
- PMCID: PMC7235778
- DOI: 10.3390/medicines7040018
Methylxanthines Inhibit Primary Amine Oxidase and Monoamine Oxidase Activities of Human Adipose Tissue
Abstract
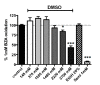
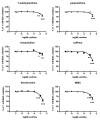
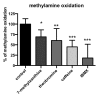
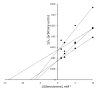
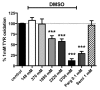
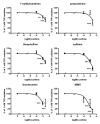
Background: Methylxanthines including caffeine and theobromine are widely consumed compounds and were recently shown to interact with bovine copper-containing amine oxidase. To the best of our knowledge, no direct demonstration of any interplay between these phytochemicals and human primary amine oxidase (PrAO) has been reported to date. We took advantage of the coexistence of PrAO and monoamine oxidase (MAO) activities in human subcutaneous adipose tissue (hScAT) to test the interaction between several methylxanthines and these enzymes, which are involved in many key pathophysiological processes. Methods: Benzylamine, methylamine, and tyramine were used as substrates for PrAO and MAO in homogenates of subcutaneous adipose depots obtained from overweight women undergoing plastic surgery. Methylxanthines were tested as substrates or inhibitors by fluorimetric determination of hydrogen peroxide, an end-product of amine oxidation. Results: Semicarbazide-sensitive PrAO activity was inhibited by theobromine, caffeine, and isobutylmethylxanthine (IBMX) while theophylline, paraxanthine, and 7-methylxanthine had little effect. Theobromine inhibited PrAO activity by 54% at 2.5 mM. Overall, the relationship between methylxanthine structure and the degree of inhibition was similar to that seen with bovine PrAO, although higher concentrations (mM) were required for inhibition. Theobromine also inhibited oxidation of tyramine by MAO, at the limits of its solubility in a DMSO vehicle. At doses higher than 12 % v/v, DMSO impaired MAO activity. MAO was also inhibited by millimolar doses of IBMX, caffeine and by other methylxanthines to a lesser extent. Conclusions: This preclinical study extrapolates previous findings with bovine PrAO to human tissues. Given that PrAO is a potential target for anti-inflammatory drugs, it indicates that alongside phosphodiesterase inhibition and adenosine receptor antagonism, PrAO and MAO inhibition could contribute to the health benefits of methylxanthines, especially their anti-inflammatory effects.
Keywords: DMSO; adipose tissue; caffeine; copper-containing amine oxidase; hydrogen peroxide; methylxanthine; monoamine oxidase; semicarbazide; theobromine.
Conflict of interest statement
The authors declare that there are no conflict of interest.
Figures
References
-
- Chen J.F., Chern Y. Impacts of methylxanthines and adenosine receptors on neurodegeneration: Human and experimental studies. Handb. Exp. Pharmacol. 2011;200:267–310. - PubMed
LinkOut - more resources
Full Text Sources

