Mutations in penicillin-binding protein 2 from cephalosporin-resistant Neisseria gonorrhoeae hinder ceftriaxone acylation by restricting protein dynamics
- PMID: 32253235
- PMCID: PMC7247294
- DOI: 10.1074/jbc.RA120.012617
Mutations in penicillin-binding protein 2 from cephalosporin-resistant Neisseria gonorrhoeae hinder ceftriaxone acylation by restricting protein dynamics
Abstract
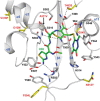
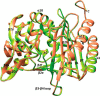
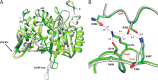
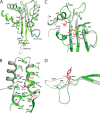
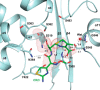
The global incidence of the sexually transmitted disease gonorrhea is expected to rise due to the spread of Neisseria gonorrhoeae strains with decreased susceptibility to extended-spectrum cephalosporins (ESCs). ESC resistance is conferred by mosaic variants of penicillin-binding protein 2 (PBP2) that have diminished capacity to form acylated adducts with cephalosporins. To elucidate the molecular mechanisms of ESC resistance, we conducted a biochemical and high-resolution structural analysis of PBP2 variants derived from the decreased-susceptibility N. gonorrhoeae strain 35/02 and ESC-resistant strain H041. Our data reveal that mutations both lower affinity of PBP2 for ceftriaxone and restrict conformational changes that normally accompany acylation. Specifically, we observe that a G545S substitution hinders rotation of the β3 strand necessary to form the oxyanion hole for acylation and also traps ceftriaxone in a noncanonical configuration. In addition, F504L and N512Y substitutions appear to prevent bending of the β3-β4 loop that is required to contact the R1 group of ceftriaxone in the active site. Other mutations also appear to act by reducing flexibility in the protein. Overall, our findings reveal that restriction of protein dynamics in PBP2 underpins the ESC resistance of N. gonorrhoeae.
Keywords: Neisseria gonorrhoeae; X-ray crystallography; antibiotic resistance; ceftriaxone; conformational change; extended-spectrum cephalosporins; oxyanion hole; penicillin-binding protein; peptidoglycan; protein dynamic.
© 2020 Singh et al.
Conflict of interest statement
The authors declare that they have no conflicts of interest with the contents of this article
Figures

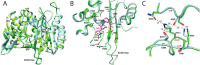
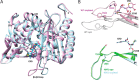

References
-
- Unemo M., Lahra M. M., Cole M., Galarza P., Ndowa F., Martin I., Dillon J. R., Ramon-Pardo P., Bolan G., and Wi T.. 2019) World Health Organization Global Gonococcal Antimicrobial Surveillance Program (WHO GASP): review of new data and evidence to inform international collaborative actions and research efforts. Sex. Health 16, 412–425 10.1071/SH19023 - DOI - PMC - PubMed
-
- Public Health Agency of Canada. 2013) Canadian Guidelines on Sexually Transmitted Infections–Management and treatment of specific infections–Gonococcal Infections. (Modified Sept., 2017) Government of Canada, Ottawa, Ontario, Canada: https://www.canada.ca/en/public-health/services/infectious-diseases/sexu...
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

