Activity in grafted human iPS cell-derived cortical neurons integrated in stroke-injured rat brain regulates motor behavior
- PMID: 32253308
- PMCID: PMC7183146
- DOI: 10.1073/pnas.2000690117
Activity in grafted human iPS cell-derived cortical neurons integrated in stroke-injured rat brain regulates motor behavior
Abstract
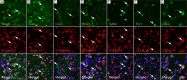
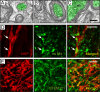
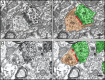
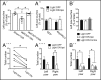
Stem cell transplantation can improve behavioral recovery after stroke in animal models but whether stem cell-derived neurons become functionally integrated into stroke-injured brain circuitry is poorly understood. Here we show that intracortically grafted human induced pluripotent stem (iPS) cell-derived cortical neurons send widespread axonal projections to both hemispheres of rats with ischemic lesions in the cerebral cortex. Using rabies virus-based transsynaptic tracing, we find that at 6 mo after transplantation, host neurons in the contralateral somatosensory cortex receive monosynaptic inputs from grafted neurons. Immunoelectron microscopy demonstrates myelination of the graft-derived axons in the corpus callosum and that their terminals form excitatory, glutamatergic synapses on host cortical neurons. We show that the stroke-induced asymmetry in a sensorimotor (cylinder) test is reversed by transplantation. Light-induced inhibition of halorhodopsin-expressing, grafted neurons does not recreate the impairment, indicating that its reversal is not due to neuronal activity in the graft. However, we find bilateral decrease of motor performance in the cylinder test after light-induced inhibition of either grafted or endogenous halorhodopsin-expressing cortical neurons, located in the same area, and after inhibition of endogenous halorhodopsin-expressing cortical neurons by exposure of their axons to light on the contralateral side. Our data indicate that activity in the grafted neurons, probably mediated through transcallosal connections to the contralateral hemisphere, is involved in maintaining normal motor function. This is an example of functional integration of efferent projections from grafted neurons into the stroke-affected brain's neural circuitry, which raises the possibility that such repair might be achievable also in humans affected by stroke.
Keywords: cerebral cortex; iPS cells; optogenetics; stroke; transplantation.
Conflict of interest statement
The authors declare no competing interest.
Figures



References
-
- Hess D. C., et al. , Safety and efficacy of multipotent adult progenitor cells in acute ischaemic stroke (MASTERS): A randomised, double-blind, placebo-controlled, phase 2 trial. Lancet Neurol. 16, 360–368 (2017). - PubMed
-
- Kalladka D., et al. , Human neural stem cells in patients with chronic ischaemic stroke (PISCES): A phase 1, first-in-man study. Lancet 388, 787–796 (2016). - PubMed
-
- Kokaia Z., Martino G., Schwartz M., Lindvall O., Cross-talk between neural stem cells and immune cells: The key to better brain repair? Nat. Neurosci. 15, 1078–1087 (2012). - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

