Effect of TLR2 on the proliferation of inflammation-related colorectal cancer and sporadic colorectal cancer
- PMID: 32256204
- PMCID: PMC7104506
- DOI: 10.1186/s12935-020-01184-0
Effect of TLR2 on the proliferation of inflammation-related colorectal cancer and sporadic colorectal cancer
Abstract
Background: Colitis-associated cancer (CAC) is a complication of inflammatory bowel disease (IBD) with a poor prognosis because it is often diagnosed in advanced stages with local progression or metastasis. Compared with the more common polyp-induced sporadic colorectal cancer (sCRC), CAC has different molecular mechanisms. Toll-like receptor 2 (TLR2) expression is not limited to cells related to inflammation and immune function. High levels of TLR2 expression in tumor tissues of colorectal cancer (CRC) patients have been reported. This report is to investigate the effects of knockout and knockdown of the TLR2 gene on the proliferation of CAC and sCRC.
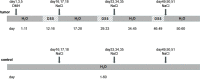
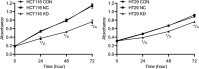
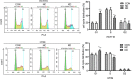
Methods: Twelve C57BL/6 J wild-type mice (WT) and 12 TLR2 knockout mice (TLR2-/-) were used to rapidly establish a colitis-associated cancer (CAC) model via the 1,2-dimethylhydrazine-dextran sodium sulfate (DMH-DSS) method and were divided into the normal WT control group (NC), TLR2 knockout control group (KC), normal wild-type tumor modeling group (NT), and TLR2 knockout tumor modeling group (KT), with 6 mice in each group. The general performance of the mice during modeling, the gross changes of the colon and the rectum, and the pathological score of HE staining were used to observe tumor growth. The expression of TLR2 was detected by immunohistochemistry, and tumor proliferation was detected by Ki67 labeling. Lentivirus carrying TLR2-RNAi was used to stably infect colorectal cancer cells (HCT116 and HT29) to knock down TLR2 gene expression. The experimental groups included the uninfected control group, negative control group, and gene knockdown group. After infection, the expression of TLR2 protein was detected by Western blot, and cell proliferation and the cell cycle were detected by the CCK-8 method and fluorescence-activated cell sorting. Western blot was used to detect the expression levels of p- NF-κβ, cyclin D1 and cyclin D3 protein in each group of cells.
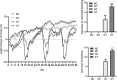
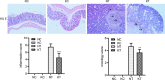
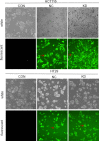
Results: TLR2 knockout in the CAC model resulted in greater changes in body weight and more severe diarrhea and colorectal hemorrhage. However, knocking out the TLR2 gene reduced the shortening of colorectal length, the number of tumors, and the total tumor volume and inhibited the growth of CAC. Knocking out the TLR2 gene also reduced the pathological score and tumor severity. TLR2 was localized in the cell membrane of the colorectal epithelium of the NC group and of the colorectal tumors of the NT group and was highly expressed in the NT group, while antigen Ki67 was localized in the nucleus of the colorectal tumor cells of the NT group and the KT group, and its expression was reduced in the KT group. In an in vitro sporadic colorectal cancer cell experiment, TLR2 protein in the TLR2 knockdown group was significantly downregulated, and TLR2 knockdown significantly inhibited the proliferation of HCT116 and HT29 colorectal cancer cells, resulting in G1 phase arrest. The expression levels of p-NF-κβ, cyclin D1 and cyclin D3 proteins in TLR2 gene knockdown group cells were significantly reduced.
Conclusion: Knockout and knockdown of TLR2 can inhibit the proliferation of inflammation-related colorectal cancer and sporadic colorectal cancer.
Keywords: Cell proliferation; Inflammation-related colorectal cancer; Sporadic colorectal cancer; Toll-like receptor 2.
© The Author(s) 2020.
Conflict of interest statement
Competing interestsThe authors declare that they have no competing interests.
Figures




Similar articles
-
Tumor Necrosis Factor Ligand-Related Molecule 1A Regulates the Occurrence of Colitis-Associated Colorectal Cancer.Dig Dis Sci. 2018 Sep;63(9):2341-2350. doi: 10.1007/s10620-018-5126-0. Epub 2018 May 24. Dig Dis Sci. 2018. PMID: 29796912
-
MicroRNA 301A Promotes Intestinal Inflammation and Colitis-Associated Cancer Development by Inhibiting BTG1.Gastroenterology. 2017 May;152(6):1434-1448.e15. doi: 10.1053/j.gastro.2017.01.049. Epub 2017 Feb 11. Gastroenterology. 2017. PMID: 28193514
-
PIAS3-mediated feedback loops promote chronic colitis-associated malignant transformation.Theranostics. 2018 Apr 30;8(11):3022-3037. doi: 10.7150/thno.23046. eCollection 2018. Theranostics. 2018. PMID: 29896300 Free PMC article.
-
Molecular mechanisms in colitis-associated colorectal cancer.Oncogenesis. 2023 Oct 26;12(1):48. doi: 10.1038/s41389-023-00492-0. Oncogenesis. 2023. PMID: 37884500 Free PMC article. Review.
-
Colorectal cancer: genetic abnormalities, tumor progression, tumor heterogeneity, clonal evolution and tumor-initiating cells.Med Sci (Basel). 2018 Apr 13;6(2):31. doi: 10.3390/medsci6020031. Med Sci (Basel). 2018. PMID: 29652830 Free PMC article. Review.
Cited by
-
A Series of Genes for Predicting Responses to Anti-Tumor Necrosis Factor α Therapy in Crohn's Disease.Front Pharmacol. 2022 Apr 20;13:870796. doi: 10.3389/fphar.2022.870796. eCollection 2022. Front Pharmacol. 2022. PMID: 35517818 Free PMC article.
-
Pattern recognition receptors in health and diseases.Signal Transduct Target Ther. 2021 Aug 4;6(1):291. doi: 10.1038/s41392-021-00687-0. Signal Transduct Target Ther. 2021. PMID: 34344870 Free PMC article. Review.
-
The Association Between Gut Microbiota, Toll-Like Receptors, and Colorectal Cancer.Clin Med Insights Oncol. 2022 Nov 1;16:11795549221130549. doi: 10.1177/11795549221130549. eCollection 2022. Clin Med Insights Oncol. 2022. PMID: 36338264 Free PMC article. Review.
-
o-Aminoazotoluene, 7,12-dimethylbenz[a]anthracene, and N-ethyl-N-nitrosourea, which are mutagenic but not carcinogenic in the colon, rapidly induce colonic tumors in mice with dextran sulfate sodium-induced colitis.Genes Environ. 2022 Mar 29;44(1):11. doi: 10.1186/s41021-022-00240-7. Genes Environ. 2022. PMID: 35351212 Free PMC article.
-
Toll-like receptor-guided therapeutic intervention of human cancers: molecular and immunological perspectives.Front Immunol. 2023 Sep 26;14:1244345. doi: 10.3389/fimmu.2023.1244345. eCollection 2023. Front Immunol. 2023. PMID: 37822929 Free PMC article. Review.
References
LinkOut - more resources
Full Text Sources
Research Materials

