Perirenal Adipose Tissue Displays an Age-Dependent Inflammatory Signature Associated With Early Graft Dysfunction of Marginal Kidney Transplants
- PMID: 32256495
- PMCID: PMC7089962
- DOI: 10.3389/fimmu.2020.00445
Perirenal Adipose Tissue Displays an Age-Dependent Inflammatory Signature Associated With Early Graft Dysfunction of Marginal Kidney Transplants
Abstract
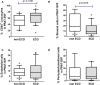
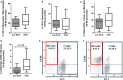
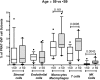
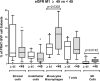
Background: Better understanding of the contribution of donor aging and comorbidity factors of expanded criteria donors (ECD) to the clinical outcome of a transplant is a challenge in kidney transplantation. We investigated whether the features of donor-derived stromal vascular fraction of perirenal adipose tissue (PRAT-SVF) could be indicative of the deleterious impact of the ECD microenvironment on a renal transplant. Methods: A comparative analysis of cellular components, transcriptomic and vasculogenic profiles was performed in PRAT-SVF obtained from 22 optimal donors and 31 ECD deceased donors. We then investigated whether these parameters could be associated with donor aging and early allograft dysfunction. Results: When compared with the PRAT-SVF of non-ECD donors, ECD PRAT-SVF displayed a lower proportion of stromal cells, a higher proportion of inflammatory NK cells. The global RNA sequencing approach indicated a differential molecular signature in the PRAT-SVF of ECD donors characterized by the over-expression of CXCL1 and IL1-β inflammatory transcripts. The vasculogenic activity of PRAT-SVF was highly variable but was not significantly affected in marginal donors. Periorgan recruitment of monocytes/macrophages and NK cells in PRAT-SVF was associated with donor aging. The presence of NK cell infiltrates was associated with lower PRAT-SVF angiogenic activity and with early allograft dysfunction evaluated on day 7 and at 1 month post-transplant. Conclusions: Our results indicate that human NK cell subsets are differentially recruited in the periorgan environment of aging kidney transplants. We provide novel evidence that PRAT-SVF represents a non-invasive and timely source of donor material with potential value to assess inflammatory features that impact organ quality and function.
Keywords: endothelial inflammation; kidney allograft dysfunction; kidney transplantation; marginal kidney donors; natural killer cells; perirenal adipose tissue.
Copyright © 2020 Boissier, François, Gondran Tellier, Meunier, Lyonnet, Simoncini, Magalon, Legris, Arnaud, Giraudo, Dignat George, Karsenty, Burtey, Lechevallier, Sabatier and Paul.
Figures


Similar articles
-
Perirenal Adipose Tissue Inflammation: Novel Insights Linking Metabolic Dysfunction to Renal Diseases.Front Endocrinol (Lausanne). 2021 Aug 2;12:707126. doi: 10.3389/fendo.2021.707126. eCollection 2021. Front Endocrinol (Lausanne). 2021. PMID: 34408726 Free PMC article. Review.
-
Depot-specific metabolic and inflammatory profiles in perirenal and renal sinus adipose tissue.Mol Med. 2025 Jul 22;31(1):262. doi: 10.1186/s10020-025-01323-1. Mol Med. 2025. PMID: 40696281 Free PMC article.
-
Impact of acute kidney injury in expanded criteria deceased donors on post-transplant clinical outcomes: multicenter cohort study.BMC Nephrol. 2019 Feb 4;20(1):39. doi: 10.1186/s12882-019-1225-1. BMC Nephrol. 2019. PMID: 30717699 Free PMC article.
-
Molecular Fingerprints of Borderline Changes in Kidney Allografts Are Influenced by Donor Category.Front Immunol. 2020 Mar 25;11:423. doi: 10.3389/fimmu.2020.00423. eCollection 2020. Front Immunol. 2020. PMID: 32269565 Free PMC article.
-
The role of perirenal adipose tissue deposition in chronic kidney disease progression: Mechanisms and therapeutic implications.Life Sci. 2024 Sep 1;352:122866. doi: 10.1016/j.lfs.2024.122866. Epub 2024 Jun 25. Life Sci. 2024. PMID: 38936605 Review.
Cited by
-
Perirenal Adipose Tissue-Current Knowledge and Future Opportunities.J Clin Med. 2021 Mar 21;10(6):1291. doi: 10.3390/jcm10061291. J Clin Med. 2021. PMID: 33800984 Free PMC article. Review.
-
Perirenal Adipose Tissue Inflammation: Novel Insights Linking Metabolic Dysfunction to Renal Diseases.Front Endocrinol (Lausanne). 2021 Aug 2;12:707126. doi: 10.3389/fendo.2021.707126. eCollection 2021. Front Endocrinol (Lausanne). 2021. PMID: 34408726 Free PMC article. Review.
-
An enhanced level of VCAM in transplant preservation fluid is an independent predictor of early kidney allograft dysfunction.Front Immunol. 2022 Aug 11;13:966951. doi: 10.3389/fimmu.2022.966951. eCollection 2022. Front Immunol. 2022. PMID: 36032101 Free PMC article.
-
Involvement of Inflammasome Components in Kidney Disease.Antioxidants (Basel). 2022 Jan 27;11(2):246. doi: 10.3390/antiox11020246. Antioxidants (Basel). 2022. PMID: 35204131 Free PMC article. Review.
-
A Review of Current and Emerging Trends in Donor Graft-Quality Assessment Techniques.J Clin Med. 2022 Jan 18;11(3):487. doi: 10.3390/jcm11030487. J Clin Med. 2022. PMID: 35159939 Free PMC article. Review.
References
-
- Ojo AO, Hanson JA, Meier-Kriesche H, Okechukwu CN, Wolfe RA, Leichtman AB, et al. . Survival in recipients of marginal cadaveric donor kidneys compared with other recipients and wait-listed transplant candidates. J Am Soc Nephrol. (2001) 12:589–97. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

