Skeletal Muscle Is an Antigen Reservoir in Integrase-Defective Lentiviral Vector-Induced Long-Term Immunity
- PMID: 32258216
- PMCID: PMC7114633
- DOI: 10.1016/j.omtm.2020.03.008
Skeletal Muscle Is an Antigen Reservoir in Integrase-Defective Lentiviral Vector-Induced Long-Term Immunity
Abstract
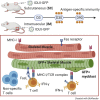
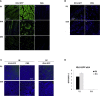
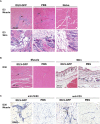
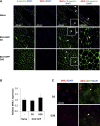
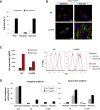
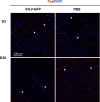
We previously developed integrase-defective lentiviral vectors (IDLVs) as an antigen delivery system for inducing strong and prolonged immunity in animal models. Here, we examined the association between persistence of antigen expression and durability of immune response. Following a single intramuscular (i.m.) or subcutaneous (s.c.) injection of IDLV delivering GFP in mice, we evaluated antigen expression and inflammation at the site of injection and persistence of antigen-specific T cells at early and late time points. Durable antigen expression was detected up to 90 days only after i.m. immunization. Mononuclear inflammation was evident soon after IDLV injection in both i.m. and s.c. immunized mice, but remained detectable up to 30 days postinjection only in i.m. immunized mice. Similarly, GFP-specific T cells were more persistent in the i.m. immunized mice. Interestingly, GFP+ muscle fibers were co-expressing major histocompatibility complex (MHC) class I, suggesting that muscle cells are competent for presenting antigens to T cells in vivo. In in vitro experiments, we demonstrated that although both primary myoblasts and myocytes present the antigen to GFP-specific T cells through MHC class I, myoblasts are more resistant to Fas-dependent cytotoxic T lymphocyte (CTL) killing activity. Overall, these data indicate that muscle cells may serve as an antigen reservoir that contributes to the long-term immunity induced by IDLV vaccination.
Keywords: CTL; Fas; GFP; MHC I; integrase defective lentiviral vectors; intramuscular immunization; muscle; myoblasts; myocytes; vaccine.
© 2020 The Author(s).
Figures



Similar articles
-
Integrase Defective Lentiviral Vector as a Vaccine Platform for Delivering Influenza Antigens.Front Immunol. 2018 Feb 5;9:171. doi: 10.3389/fimmu.2018.00171. eCollection 2018. Front Immunol. 2018. PMID: 29459873 Free PMC article.
-
Integrase Defective Lentiviral Vector Promoter Impacts Transgene Expression in Target Cells and Magnitude of Vector-Induced Immune Responses.Viruses. 2023 Nov 14;15(11):2255. doi: 10.3390/v15112255. Viruses. 2023. PMID: 38005931 Free PMC article.
-
Optimization of mucosal responses after intramuscular immunization with integrase defective lentiviral vector.PLoS One. 2014 Sep 11;9(9):e107377. doi: 10.1371/journal.pone.0107377. eCollection 2014. PLoS One. 2014. PMID: 25210766 Free PMC article.
-
Integrase-defective lentiviral-vector-based vaccine: a new vector for induction of T cell immunity.Expert Opin Biol Ther. 2011 Jun;11(6):739-50. doi: 10.1517/14712598.2011.571670. Epub 2011 Mar 25. Expert Opin Biol Ther. 2011. PMID: 21434847 Review.
-
DNA-mediated immunization to the hepatitis B surface antigen. Activation and entrainment of the immune response.Ann N Y Acad Sci. 1995 Nov 27;772:64-76. doi: 10.1111/j.1749-6632.1995.tb44732.x. Ann N Y Acad Sci. 1995. PMID: 8546414 Review.
Cited by
-
Integrase-Defective Lentiviral Vector Is an Efficient Vaccine Platform for Cancer Immunotherapy.Viruses. 2021 Feb 23;13(2):355. doi: 10.3390/v13020355. Viruses. 2021. PMID: 33672349 Free PMC article.
-
Safety and efficiency modifications of SIV-based integrase-defective lentiviral vectors for immunization.Mol Ther Methods Clin Dev. 2021 Oct 1;23:263-275. doi: 10.1016/j.omtm.2021.09.011. eCollection 2021 Dec 10. Mol Ther Methods Clin Dev. 2021. PMID: 34729374 Free PMC article.
-
Integrase-Defective Lentiviral Vectors for Delivery of Monoclonal Antibodies against Influenza.Viruses. 2020 Dec 17;12(12):1460. doi: 10.3390/v12121460. Viruses. 2020. PMID: 33348840 Free PMC article.
-
Persistent immunogenicity of integrase defective lentiviral vectors delivering membrane-tethered native-like HIV-1 envelope trimers.NPJ Vaccines. 2022 Apr 21;7(1):44. doi: 10.1038/s41541-022-00465-1. NPJ Vaccines. 2022. PMID: 35449174 Free PMC article.
-
Adeno-associated virus-vectored delivery of HIV biologics: the promise of a "single-shot" functional cure for HIV infection.J Virus Erad. 2023 Feb 17;9(1):100316. doi: 10.1016/j.jve.2023.100316. eCollection 2023 Mar. J Virus Erad. 2023. PMID: 36915910 Free PMC article.
References
-
- Liu M.A. Gene-based vaccines: Recent developments. Curr. Opin. Mol. Ther. 2010;12:86–93. - PubMed
-
- Ewer K.J., Lambe T., Rollier C.S., Spencer A.J., Hill A.V., Dorrell L. Viral vectors as vaccine platforms: from immunogenicity to impact. Curr. Opin. Immunol. 2016;41:47–54. - PubMed
-
- Rollier C.S., Reyes-Sandoval A., Cottingham M.G., Ewer K., Hill A.V. Viral vectors as vaccine platforms: deployment in sight. Curr. Opin. Immunol. 2011;23:377–382. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

