Downregulation of LncRNA NORAD promotes Ox-LDL-induced vascular endothelial cell injury and atherosclerosis
- PMID: 32267831
- PMCID: PMC7185106
- DOI: 10.18632/aging.103034
Downregulation of LncRNA NORAD promotes Ox-LDL-induced vascular endothelial cell injury and atherosclerosis
Abstract
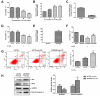
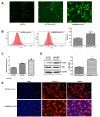
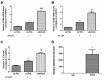
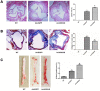
Long noncoding RNAs (lncRNAs) play important roles in the development of vascular diseases. However, the effect of lncRNA NORAD on atherosclerosis remains unknown. This study aimed to investigate the effect NORAD on endothelial cell injury and atherosclerosis. Ox-LDL-treated human umbilical vein endothelial cells (HUVECs) and high-fat-diet (HFD)-fed ApoE-/- mice were used as in vitro and in vivo models. Results showed that NORAD-knockdown induced cell cycle arrest in G0/G1 phase, aggravated ox-LDL-induced cell viability reduction, cell apoptosis, and cell senescence along with the increased expression of Bax, P53, P21 and cleaved caspase-3 and the decreased expression of Bcl-2. The effect of NORAD on cell viability was further verified via NORAD-overexpression. NORAD- knockdown increased ox-LDL-induced reactive oxygen species, malondialdehyde, p-IKBα expression levels and NF-κB nuclear translocation. Proinflammatory molecules ICAM, VCAM, and IL-8 were also increased by NORAD- knockdown. Additionally, we identified the strong interaction of NORAD and IL-8 transcription repressor SFPQ in HUVECs. In ApoE-/- mice, NORAD-knockdown increased the lipid disorder and atherosclerotic lesions. The results have suggested that lncRNA NORAD attenuates endothelial cell senescence, endothelial cell apoptosis, and atherosclerosis via NF-κB and p53-p21 signaling pathways and IL-8, in which NORAD-mediated effect on IL-8 might through the direct interaction with SFPQ.
Keywords: IL-8; NORAD; cell apoptosis; cell senescence; ox-LDL.
Conflict of interest statement
Figures


References
-
- Suciu CF, Prete M, Ruscitti P, Favoino E, Giacomelli R, Perosa F. Oxidized low density lipoproteins: the bridge between atherosclerosis and autoimmunity. Possible implications in accelerated atherosclerosis and for immune intervention in autoimmune rheumatic disorders. Autoimmun Rev. 2018; 17:366–75. 10.1016/j.autrev.2017.11.028 - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

