CDC Recommendations for Hepatitis C Screening Among Adults - United States, 2020
- PMID: 32271723
- PMCID: PMC7147910
- DOI: 10.15585/mmwr.rr6902a1
CDC Recommendations for Hepatitis C Screening Among Adults - United States, 2020
Abstract
Hepatitis C virus (HCV) infection is a major source of morbidity and mortality in the United States. HCV is transmitted primarily through parenteral exposures to infectious blood or body fluids that contain blood, most commonly through injection drug use. No vaccine against hepatitis C exists and no effective pre- or postexposure prophylaxis is available. More than half of persons who become infected with HCV will develop chronic infection. Direct-acting antiviral treatment can result in a virologic cure in most persons with 8-12 weeks of all-oral medication regimens. This report augments (i.e., updates and summarizes) previously published recommendations from CDC regarding testing for HCV infection in the United States (Smith BD, Morgan RL, Beckett GA, et al. Recommendations for the identification of chronic hepatitis C virus infection among persons born during 1945-1965. MMWR Recomm Rec 2012;61[No. RR-4]). CDC is augmenting previous guidance with two new recommendations: 1) hepatitis C screening at least once in a lifetime for all adults aged ≥18 years, except in settings where the prevalence of HCV infection is <0.1% and 2) hepatitis C screening for all pregnant women during each pregnancy, except in settings where the prevalence of HCV infection is <0.1%. The recommendation for HCV testing that remains unchanged is regardless of age or setting prevalence, all persons with risk factors should be tested for hepatitis C, with periodic testing while risk factors persist. Any person who requests hepatitis C testing should receive it, regardless of disclosure of risk, because many persons might be reluctant to disclose stigmatizing risks.
Conflict of interest statement
All authors have completed and submitted the International Committee of Medical Journal Editors form for disclosure of potential conflicts of interest. No potential conflicts of interest were disclosed.
Figures
References
-
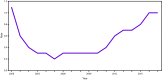
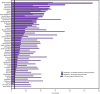
- CDC. Viral hepatitis surveillance—United States, 2017. Atlanta, GA: US Department of Health and Human Services, CDC; 2019. https://www.cdc.gov/hepatitis/statistics/2017surveillance/index.htm
-
- Smith BD, Morgan RL, Beckett GA, et al. Recommendations for the identification of chronic hepatitis C virus infection among persons born during 1945-1965. MMWR Recomm Rep 2012;61(RR-4):1–32. - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
