Disruption of the Plasmodium falciparum Life Cycle through Transcriptional Reprogramming by Inhibitors of Jumonji Demethylases
- PMID: 32272012
- PMCID: PMC7748244
- DOI: 10.1021/acsinfecdis.9b00455
Disruption of the Plasmodium falciparum Life Cycle through Transcriptional Reprogramming by Inhibitors of Jumonji Demethylases
Abstract
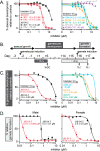
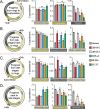
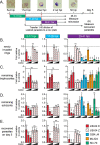
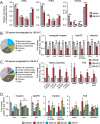
Little is known about the role of the three Jumonji C (JmjC) enzymes in Plasmodium falciparum (Pf). Here, we show that JIB-04 and other established inhibitors of mammalian JmjC histone demethylases kill asexual blood stage parasites and are even more potent at blocking gametocyte development and gamete formation. In late stage parasites, JIB-04 increased levels of trimethylated lysine residues on histones, suggesting the inhibition of P. falciparum Jumonji demethylase activity. These epigenetic defects coincide with deregulation of invasion, cell motor, and sexual development gene programs, including gene targets coregulated by the PfAP2-I transcription factor and chromatin-binding factor, PfBDP1. Mechanistically, we demonstrate that PfJmj3 converts 2-oxoglutarate to succinate in an iron-dependent manner consistent with mammalian Jumonji enzymes, and this catalytic activity is inhibited by JIB-04 and other Jumonji inhibitors. Our pharmacological studies of Jumonji activity in the malaria parasite provide evidence that inhibition of these enzymatic activities is detrimental to the parasite.
Keywords: JIB-04; Jumonji inhibitors; Plasmodium falciparum; demethylases; gametocytes; malaria; transcriptional reprogramming.
Conflict of interest statement
The authors declare no competing financial interest.
Figures

Similar articles
-
A small molecule modulates Jumonji histone demethylase activity and selectively inhibits cancer growth.Nat Commun. 2013;4:2035. doi: 10.1038/ncomms3035. Nat Commun. 2013. PMID: 23792809 Free PMC article.
-
Inhibition of Jumonji demethylases reprograms severe dilated cardiomyopathy and prolongs survival.J Biol Chem. 2022 Feb;298(2):101515. doi: 10.1016/j.jbc.2021.101515. Epub 2021 Dec 18. J Biol Chem. 2022. PMID: 34933013 Free PMC article.
-
A comprehensive study of epigenetic alterations in hepatocellular carcinoma identifies potential therapeutic targets.J Hepatol. 2019 Jul;71(1):78-90. doi: 10.1016/j.jhep.2019.03.007. Epub 2019 Mar 15. J Hepatol. 2019. PMID: 30880225
-
Structure-function relationships in KDM7 histone demethylases.Adv Protein Chem Struct Biol. 2019;117:113-125. doi: 10.1016/bs.apcsb.2019.08.005. Epub 2019 Sep 10. Adv Protein Chem Struct Biol. 2019. PMID: 31564306 Review.
-
Development of JmjC-domain-containing histone demethylase (KDM2-7) inhibitors for cancer therapy.Drug Discov Today. 2023 May;28(5):103519. doi: 10.1016/j.drudis.2023.103519. Epub 2023 Feb 6. Drug Discov Today. 2023. PMID: 36754142 Review.
Cited by
-
Genetic complexity alters drug susceptibility of asexual and gametocyte stages of Plasmodium falciparum to antimalarial candidates.Antimicrob Agents Chemother. 2024 Mar 6;68(3):e0129123. doi: 10.1128/aac.01291-23. Epub 2024 Jan 23. Antimicrob Agents Chemother. 2024. PMID: 38259087 Free PMC article.
-
Transmission-Blocking Strategies for Malaria Eradication: Recent Advances in Small-Molecule Drug Development.Pharmaceuticals (Basel). 2024 Jul 19;17(7):962. doi: 10.3390/ph17070962. Pharmaceuticals (Basel). 2024. PMID: 39065810 Free PMC article. Review.
-
Adapt or Die: Targeting Unique Transmission-Stage Biology for Malaria Elimination.Front Cell Infect Microbiol. 2022 Jun 9;12:901971. doi: 10.3389/fcimb.2022.901971. eCollection 2022. Front Cell Infect Microbiol. 2022. PMID: 35755845 Free PMC article. Review.
-
Epigenetic regulation as a therapeutic target in the malaria parasite Plasmodium falciparum.Malar J. 2024 Feb 12;23(1):44. doi: 10.1186/s12936-024-04855-9. Malar J. 2024. PMID: 38347549 Free PMC article. Review.
-
New Transmission-Selective Antimalarial Agents through Hit-to-Lead Optimization of 2-([1,1'-Biphenyl]-4-carboxamido)benzoic Acid Derivatives.Chembiochem. 2022 Nov 4;23(21):e202200427. doi: 10.1002/cbic.202200427. Epub 2022 Oct 6. Chembiochem. 2022. PMID: 36106425 Free PMC article.
References
-
- WHO (2018) World malaria report 2018, WHO, Geneva.
-
- Flueck C.; Bartfai R.; Volz J.; Niederwieser I.; Salcedo-Amaya A. M.; Alako B. T.; Ehlgen F.; Ralph S. A.; Cowman A. F.; Bozdech Z.; Stunnenberg H. G.; Voss T. S. (2009) Plasmodium falciparum heterochromatin protein 1 marks genomic loci linked to phenotypic variation of exported virulence factors. PLoS Pathog. 5 (9), e1000569.10.1371/journal.ppat.1000569. - DOI - PMC - PubMed

