Neurog2 Acts as a Classical Proneural Gene in the Ventromedial Hypothalamus and Is Required for the Early Phase of Neurogenesis
- PMID: 32273485
- PMCID: PMC7189762
- DOI: 10.1523/JNEUROSCI.2610-19.2020
Neurog2 Acts as a Classical Proneural Gene in the Ventromedial Hypothalamus and Is Required for the Early Phase of Neurogenesis
Abstract
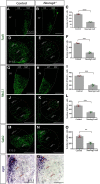
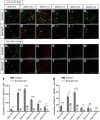
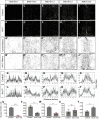
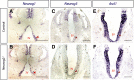
The tuberal hypothalamus is comprised of the dorsomedial, ventromedial, and arcuate nuclei, as well as parts of the lateral hypothalamic area, and it governs a wide range of physiologies. During neurogenesis, tuberal hypothalamic neurons are thought to be born in a dorsal-to-ventral and outside-in pattern, although the accuracy of this description has been questioned over the years. Moreover, the intrinsic factors that control the timing of neurogenesis in this region are poorly characterized. Proneural genes, including Achate-scute-like 1 (Ascl1) and Neurogenin 3 (Neurog3) are widely expressed in hypothalamic progenitors and contribute to lineage commitment and subtype-specific neuronal identifies, but the potential role of Neurogenin 2 (Neurog2) remains unexplored. Birthdating in male and female mice showed that tuberal hypothalamic neurogenesis begins as early as E9.5 in the lateral hypothalamic and arcuate and rapidly expands to dorsomedial and ventromedial neurons by E10.5, peaking throughout the region by E11.5. We confirmed an outside-in trend, except for neurons born at E9.5, and uncovered a rostrocaudal progression but did not confirm a dorsal-ventral patterning to tuberal hypothalamic neuronal birth. In the absence of Neurog2, neurogenesis stalls, with a significant reduction in early-born BrdU+ cells but no change at later time points. Further, the loss of Ascl1 yielded a similar delay in neuronal birth, suggesting that Ascl1 cannot rescue the loss of Neurog2 and that these proneural genes act independently in the tuberal hypothalamus. Together, our findings show that Neurog2 functions as a classical proneural gene to regulate the temporal progression of tuberal hypothalamic neurogenesis.SIGNIFICANCE STATEMENT Here, we investigated the general timing and pattern of neurogenesis within the tuberal hypothalamus. Our results confirmed an outside-in trend of neurogenesis and uncovered a rostrocaudal progression. We also showed that Neurog2 acts as a classical proneural gene and is responsible for regulating the birth of early-born neurons within the ventromedial hypothalamus, acting independently of Ascl1 In addition, we revealed a role for Neurog2 in cell fate specification and differentiation of ventromedial -specific neurons. Last, Neurog2 does not have cross-inhibitory effects on Neurog1, Neurog3, and Ascl1 These findings are the first to reveal a role for Neurog2 in hypothalamic development.
Keywords: Neurog2; VMH; neurogenesis; proneural genes; specification.
Copyright © 2020 the authors.
Figures
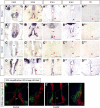
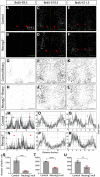
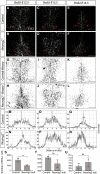
Similar articles
-
Neurog2 and Ascl1 together regulate a postmitotic derepression circuit to govern laminar fate specification in the murine neocortex.Proc Natl Acad Sci U S A. 2017 Jun 20;114(25):E4934-E4943. doi: 10.1073/pnas.1701495114. Epub 2017 Jun 5. Proc Natl Acad Sci U S A. 2017. PMID: 28584103 Free PMC article.
-
Ascl1 is required to specify a subset of ventromedial hypothalamic neurons.Development. 2020 May 28;147(10):dev180067. doi: 10.1242/dev.180067. Development. 2020. PMID: 32253239
-
GSK3 temporally regulates neurogenin 2 proneural activity in the neocortex.J Neurosci. 2012 Jun 6;32(23):7791-805. doi: 10.1523/JNEUROSCI.1309-12.2012. J Neurosci. 2012. PMID: 22674256 Free PMC article.
-
Instructing neuronal identity during CNS development and astroglial-lineage reprogramming: Roles of NEUROG2 and ASCL1.Brain Res. 2019 Feb 15;1705:66-74. doi: 10.1016/j.brainres.2018.02.045. Epub 2018 Mar 3. Brain Res. 2019. PMID: 29510143 Review.
-
Proneural genes in neocortical development.Neuroscience. 2013 Dec 3;253:256-73. doi: 10.1016/j.neuroscience.2013.08.029. Epub 2013 Aug 30. Neuroscience. 2013. PMID: 23999125 Review.
Cited by
-
TAT-LBD-Ngn2-improved cognitive functions after global cerebral ischemia by enhancing neurogenesis.Brain Behav. 2023 Jan;13(1):e2847. doi: 10.1002/brb3.2847. Epub 2022 Dec 10. Brain Behav. 2023. PMID: 36495119 Free PMC article.
-
Single-cell analysis of early chick hypothalamic development reveals that hypothalamic cells are induced from prethalamic-like progenitors.Cell Rep. 2022 Jan 18;38(3):110251. doi: 10.1016/j.celrep.2021.110251. Cell Rep. 2022. PMID: 35045288 Free PMC article.
-
Gestational low-dose BPA exposure impacts suprachiasmatic nucleus neurogenesis and circadian activity with transgenerational effects.Sci Adv. 2021 May 28;7(22):eabd1159. doi: 10.1126/sciadv.abd1159. Print 2021 May. Sci Adv. 2021. PMID: 34049886 Free PMC article.
-
Control of tuberal hypothalamic development and its implications in metabolic disorders.Nat Rev Endocrinol. 2025 Feb;21(2):118-130. doi: 10.1038/s41574-024-01036-1. Epub 2024 Sep 23. Nat Rev Endocrinol. 2025. PMID: 39313573 Free PMC article. Review.
-
Glial Cell Reprogramming in Ischemic Stroke: A Review of Recent Advancements and Translational Challenges.Transl Stroke Res. 2025 Oct;16(5):1811-1835. doi: 10.1007/s12975-025-01331-7. Epub 2025 Feb 4. Transl Stroke Res. 2025. PMID: 39904845 Review.
References
-
- Akagi T, Inoue T, Miyoshi G, Bessho Y, Takahashi M, Lee J, Guillemot F, Kageyama R (2004) Requirement of multiple basic helix-loop-helix genes for retinal neuronal subtype specification. J Biol Chem 279:28492–28498. - PubMed
-
- Altman J, Bayer S (1986) The development of the rat hypothalamus. Adv Anat Embryol Cell Biol 100:1–178. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
