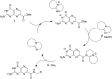
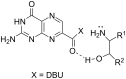
Exploring the scope of DBU-promoted amidations of 7-methoxycarbonylpterin
- PMID: 32273911
- PMCID: PMC7113547
- DOI: 10.3762/bjoc.16.46
Exploring the scope of DBU-promoted amidations of 7-methoxycarbonylpterin
Abstract
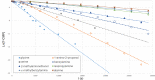
The synthetic utility of pterins is often hampered by the notorious insolubility of this heterocycle, slowing the development of medicinally relevant pteridine derivatives. Reactions which expedite the development of new pterins are thus of great importance. Through a dual role of diazabicycloundecene (DBU), 7-carboxymethylpterin is converted to the soluble DBU salt, with additional DBU promoting an ester-to-amide transformation. We have explored this reaction to assess its scope and identify structural features in the amines which significantly affect success, monitored the reaction kinetics using a pseudo-first order kinetics model, and further adapted the reaction conditions to allow for product formation in as little as 5 min, with yields often >80%.
Keywords: DBU; amidation; folate; pterin.
Copyright © 2020, Bockman and Pruet; licensee Beilstein-Institut.
Figures
Similar articles
-
Optimized 5-membered heterocycle-linked pterins for the inhibition of Ricin Toxin A.ACS Med Chem Lett. 2012 Jul 12;3(7):588-591. doi: 10.1021/ml300099t. Epub 2012 May 29. ACS Med Chem Lett. 2012. PMID: 23050058 Free PMC article.
-
Base-promoted direct amidation of esters: beyond the current scope and practical applications.RSC Adv. 2022 Jul 15;12(32):20555-20562. doi: 10.1039/d2ra03524c. eCollection 2022 Jul 14. RSC Adv. 2022. PMID: 35919171 Free PMC article.
-
Mechanisms of organocatalytic amidation and trans-esterification of aromatic esters as a model for the depolymerization of poly(ethylene) terephthalate.J Phys Chem A. 2012 Dec 27;116(51):12389-98. doi: 10.1021/jp304212y. Epub 2012 Dec 14. J Phys Chem A. 2012. PMID: 23241219
-
Pteridine derivatives as modulators of oxidative stress.Curr Drug Metab. 2002 Apr;3(2):203-9. doi: 10.2174/1389200024605127. Curr Drug Metab. 2002. PMID: 12003351 Review.
-
Commentary: The roles of folate and pteridine derivatives in neurotransmitter metabolism.Biochem Pharmacol. 1977 Jun 1;26(11):1009-14. doi: 10.1016/0006-2952(77)90236-2. Biochem Pharmacol. 1977. PMID: 18148 Review. No abstract available.
References
LinkOut - more resources
Full Text Sources