Oxidant/Antioxidant Profile in the Thoracic Aneurysm of Patients with the Loeys-Dietz Syndrome
- PMID: 32273946
- PMCID: PMC7128053
- DOI: 10.1155/2020/5392454
Oxidant/Antioxidant Profile in the Thoracic Aneurysm of Patients with the Loeys-Dietz Syndrome
Abstract
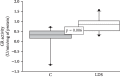
Patients with the Loeys-Dietz syndrome (LDS) have mutations in the TGF-βR1, TGF-βR2, and SMAD3 genes. However, little is known about the redox homeostasis in the thoracic aortic aneurysms (TAA) they develop. Here, we evaluate the oxidant/antioxidant profile in the TAA tissue from LDS patients and compare it with that in nondamaged aortic tissue from control (C) subjects. We evaluate the enzymatic activities of glutathione peroxidase (GPx), glutathione S-transferase (GST), glutathione reductase (GR), catalase (CAT), superoxide dismutase (SOD) isoforms, and thioredoxin reductase (TrxR). We also analyze some antioxidants from a nonenzymatic system such as selenium (Se), glutathione (GSH), and total antioxidant capacity (TAC). Oxidative stress markers such as lipid peroxidation and carbonylation, as well as xanthine oxidase (ORX) and nuclear factor erythroid 2-related factor 2 (Nrf2) expressions, were also evaluated. TAA from LDS patients showed a decrease in GSH, Se, TAC, GPx, GST, CAT, and TrxR. The SOD activity and ORX expressions were increased, but the Nrf2 expression was decreased. The results suggest that the redox homeostasis is altered in the TAA from LDS patients, favoring ROS overproduction that contributes to the decrease in GSH and TAC and leads to LPO and carbonylation. The decrease in Se and Nrf2 alters the activity and/or expression of some antioxidant enzymes, thus favoring a positive feedback oxidative background that contributes to the TAA formation.
Copyright © 2020 Maria Elena Soto et al.
Conflict of interest statement
All the authors declare that there are no competing interests regarding the publication of this paper.
Figures






References
-
- Kuzmik G., Sang A., Elefteriandes J. Natural history of thoracic aortic aneurysms. Journal of Vascular Surgery. 2012;56(2):561–575. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

