Nickel/Photoredox-Catalyzed Methylation of (Hetero)aryl Chlorides Using Trimethyl Orthoformate as a Methyl Radical Source
- PMID: 32275411
- PMCID: PMC7450724
- DOI: 10.1021/jacs.0c02805
Nickel/Photoredox-Catalyzed Methylation of (Hetero)aryl Chlorides Using Trimethyl Orthoformate as a Methyl Radical Source
Abstract
Methylation of organohalides represents a valuable transformation, but typically requires harsh reaction conditions or reagents. We report a radical approach for the methylation of (hetero)aryl chlorides using nickel/photoredox catalysis wherein trimethyl orthoformate, a common laboratory solvent, serves as a methyl source. This method permits methylation of (hetero)aryl chlorides and acyl chlorides at an early and late stage with broad functional group compatibility. Mechanistic investigations indicate that trimethyl orthoformate serves as a source of methyl radical via β-scission from a tertiary radical generated upon chlorine-mediated hydrogen atom transfer.
Figures
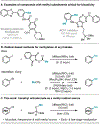
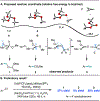


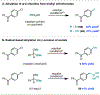
References
-
- McGrath NA; Brichacek M; Njardarson JT A Graphical Journey of Innovative Organic Architectures That Have Improved Our Lives. J. Chem. Educ 2010, 87, 1348–1349.
-
- Schönherr H; Cernak T Profound Methyl Effects in Drug Discovery and a Call for New C-H Methylation Reactions. Angew. Chem. Int. Ed 2013, 52, 12256–12267. - PubMed
-
- Yan G; Borah AJ; Wang L; Yang M Recent Advances in Transition Metal-Catalyzed Methylation Reactions. Adv. Synth. Catal 2015, 557, 1333–1350.
- Hu L; Liu YA; Liao X Recent Progress in Methylation of (Hetero)Arenes by Cross-Coupling or C-H Activation. Synlett 2018, 29, 375–382.
-
- For a recent example that overcomes many existing limitations through the use of a new methylating agent, see: He Z-T; Li H; Haydl AM; Whiteker GT; Hartwig JF Trimethylphosphate as a Methylating Agent for Cross Coupling: A Slow-Release Mechanism for the Methylation of Arylboronic Esters. J. Am. Chem. Soc 2018, 140, 17197–17202. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

