Achieving Life through Death: Redox Biology of Lipid Peroxidation in Ferroptosis
- PMID: 32275865
- PMCID: PMC7218794
- DOI: 10.1016/j.chembiol.2020.03.014
Achieving Life through Death: Redox Biology of Lipid Peroxidation in Ferroptosis
Abstract
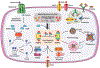
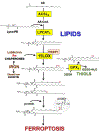
Redox balance is essential for normal brain, hence dis-coordinated oxidative reactions leading to neuronal death, including programs of regulated death, are commonly viewed as an inevitable pathogenic penalty for acute neuro-injury and neurodegenerative diseases. Ferroptosis is one of these programs triggered by dyshomeostasis of three metabolic pillars: iron, thiols, and polyunsaturated phospholipids. This review focuses on: (1) lipid peroxidation (LPO) as the major instrument of cell demise, (2) iron as its catalytic mechanism, and (3) thiols as regulators of pro-ferroptotic signals, hydroperoxy lipids. Given the central role of LPO, we discuss the engagement of selective and specific enzymatic pathways versus random free radical chemical reactions in the context of the phospholipid substrates, their biosynthesis, intracellular location, and related oxygenating machinery as participants in ferroptotic cascades. These concepts are discussed in the light of emerging neuro-therapeutic approaches controlling intracellular production of pro-ferroptotic phospholipid signals and their non-cell-autonomous spreading, leading to ferroptosis-associated necroinflammation.
Keywords: cerebral hemorrhage; cerebral ischemia; glutathione peroxidase 4; lipoxygenase; neurodegeneration; phospholipid; redox lipidomics; regulated cell death; traumatic brain injury.
Copyright © 2020 Elsevier Ltd. All rights reserved.
Conflict of interest statement
Declaration of Interests
The authors declare no competing interests.
Figures
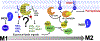
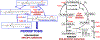



References
-
- Alim I, Caulfield JT, Chen Y, Swarup V, Geschwind DH, Ivanova E, Seravalli J, Ai Y, Sansing LH, Ste Marie EJ, et al. (2019). Selenium Drives a Transcriptional Adaptive Program to Block Ferroptosis and Treat Stroke. Cell 177, 1262–1279 e1225. - PubMed
-
- Anderson ME, Powrie F, Puri RN, and Meister A (1985). Glutathione monoethyl ester: preparation, uptake by tissues, and conversion to glutathione. Archives of biochemistry and biophysics 239, 538–548. - PubMed
-
- Angeli JPF, Shah R, Pratt DA, and Conrad M (2017). Ferroptosis Inhibition: Mechanisms and Opportunities. Trends in Pharmacological Sciences 38, 489–498. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

