Vav1 and mutant K-Ras synergize in the early development of pancreatic ductal adenocarcinoma in mice
- PMID: 32277014
- PMCID: PMC7156281
- DOI: 10.26508/lsa.202000661
Vav1 and mutant K-Ras synergize in the early development of pancreatic ductal adenocarcinoma in mice
Abstract
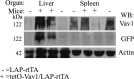
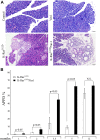
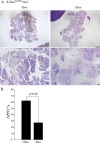
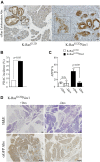
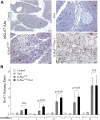
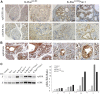
To explore the contribution of Vav1, a hematopoietic signal transducer, to pancreatic ductal adenocarcinoma (PDAC) development, we generated transgenic mouse lines expressing, Vav1, K-RasG12D, or both K-RasG12D and Vav1 in pancreatic acinar cells. Co-expression of Vav1 and K-RasG12D synergistically enhanced acinar-to-ductal metaplasia (ADM) formation, far exceeding the number of lesions developed in K-RasG12D mice. Mice expressing only Vav1 did not develop ADM. Moreover, the incidence of PDAC in K-RasG12D/Vav1 was significantly higher than in K-RasG12D mice. Discontinuing Vav1 expression in K-RasG12D/Vav1 mice elicited a marked regression of malignant lesions in the pancreas, demonstrating Vav1 is required for generation and maintenance of ADM. Rac1-GTP levels in the K-RasG12D/Vav1 mice pancreas clearly demonstrated an increase in Rac1 activity. Treatment of K-RasG12D and K-RasG12D/Vav1 mice with azathioprine, an immune-suppressor drug which inhibits Vav1's activity as a GDP/GTP exchange factor, dramatically reduced the number of malignant lesions. These results suggest that Vav1 plays a role in the development of PDAC when co-expressed with K-RasG12D via its activity as a GEF for Rac1GTPase.
© 2020 Salaymeh et al.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures





References
-
- Abate F, da Silva-Almeida AC, Zairis S, Robles-Valero J, Couronne L, Khiabanian H, Quinn SA, Kim MY, Laginestra MA, Kim C, et al. (2017) Activating mutations and translocations in the guanine exchange factor VAV1 in peripheral T-cell lymphomas. Proc Natl Acad Sci U S A 114: 764–769. 10.1073/pnas.1608839114 - DOI - PMC - PubMed
-
- Aichler M, Seiler C, Tost M, Siveke J, Mazur PK, Da Silva-Buttkus P, Bartsch DK, Langer P, Chiblak S, Durr A, et al. (2012) Origin of pancreatic ductal adenocarcinoma from atypical flat lesions: A comparative study in transgenic mice and human tissues. J Pathol 226: 723–734. 10.1002/path.3017 - DOI - PubMed
-
- Bardeesy N, Aguirre AJ, Chu GC, Cheng KH, Lopez LV, Hezel AF, Feng B, Brennan C, Weissleder R, Mahmood U, et al. (2006) Both p16(Ink4a) and the p19(Arf)-p53 pathway constrain progression of pancreatic adenocarcinoma in the mouse. Proc Natl Acad Sci U S A 103: 5947–5952. 10.1073/pnas.0601273103 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous
