Diagnostic Testing for Severe Acute Respiratory Syndrome-Related Coronavirus 2: A Narrative Review
- PMID: 32282894
- PMCID: PMC7170415
- DOI: 10.7326/M20-1301
Diagnostic Testing for Severe Acute Respiratory Syndrome-Related Coronavirus 2: A Narrative Review
Abstract
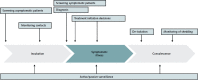
Diagnostic testing to identify persons infected with severe acute respiratory syndrome-related coronavirus 2 (SARS-CoV-2) infection is central to control the global pandemic of COVID-19 that began in late 2019. In a few countries, the use of diagnostic testing on a massive scale has been a cornerstone of successful containment strategies. In contrast, the United States, hampered by limited testing capacity, has prioritized testing for specific groups of persons. Real-time reverse transcriptase polymerase chain reaction-based assays performed in a laboratory on respiratory specimens are the reference standard for COVID-19 diagnostics. However, point-of-care technologies and serologic immunoassays are rapidly emerging. Although excellent tools exist for the diagnosis of symptomatic patients in well-equipped laboratories, important gaps remain in screening asymptomatic persons in the incubation phase, as well as in the accurate determination of live viral shedding during convalescence to inform decisions to end isolation. Many affluent countries have encountered challenges in test delivery and specimen collection that have inhibited rapid increases in testing capacity. These challenges may be even greater in low-resource settings. Urgent clinical and public health needs currently drive an unprecedented global effort to increase testing capacity for SARS-CoV-2 infection. Here, the authors review the current array of tests for SARS-CoV-2, highlight gaps in current diagnostic capacity, and propose potential solutions.
Keywords: Antibodies; Antigens; COVID-19; Cells; Infectious disease immunology; Nucleic acids; Prevention, policy, and public health; Pulmonary diseases; Research laboratories; Upper respiratory tract infections.
Figures

References
-
- World Health Organization. Novel coronavirus (COVID-19) situation. Accessed at https://experience.arcgis.com/experience/685d0ace521648f8a5beeeee1b9125cd. on 24 March 2020.
-
- Mizumoto K, Kagaya K, Chowell G. Early epidemiological assessment of the transmission potential and virulence of coronavirus disease 2019 (COVID-19) in Wuhan City: China, January-February, 2020. Accessed at https://www.medrxiv.org/content/10.1101/2020.02.12.20022434v2. on 24 March 2020.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
