Isoliquiritigenin Derivative Regulates miR-374a/BAX Axis to Suppress Triple-Negative Breast Cancer Tumorigenesis and Development
- PMID: 32296334
- PMCID: PMC7137655
- DOI: 10.3389/fphar.2020.00378
Isoliquiritigenin Derivative Regulates miR-374a/BAX Axis to Suppress Triple-Negative Breast Cancer Tumorigenesis and Development
Abstract
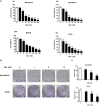
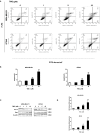
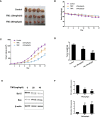
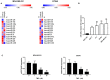
Triple-negative breast cancer (TNBC) is a subtype of breast cancer that accounts for the largest proportion of breast cancer-related deaths. Thus, it is imperative to search for novel drug candidates with potent anti-TNBC effects. Recent studies suggest that isoliquiritigenin (ISL) can significantly suppress the growth, migration, and invasion of breast cancer cells. We previously synthesized ISL derivatives and found that 3',4',5',4″-tetramethoxychalcone (TMC) inhibits TNBC cell proliferation to a greater degree than ISL. The present study aimed to investigate the mechanisms underlying the anti-TNBC effects of TMC in vitro and in vivo. We show that TMC significantly inhibits the proliferative, migratory, and invasive abilities of MDA-MB-231 and BT549 cells. TMC induces apoptosis through the upregulation of Bax and downregulation of Bcl-2. PCR arrays demonstrate a significant decrease in miR-374a expression in TNBC cells after 24-h TMC treatment. MiR-374a is overexpressed in TNBC cells and has oncogenic properties. Real-time PCR analysis confirmed that TMC inhibits miR-374a in a dose-dependent manner, and luciferase assays confirmed that BAX is targeted by miR-374a. Further, we show that TMC increases Bax protein and mRNA levels by inhibiting miR-374a. TMC also attenuates TNBC tumor volumes and weights in vivo. These results demonstrate that TMC inhibits TNBC cell proliferation, foci formation, migration, invasion, and tumorigenesis, suggesting its potential to serve as a novel drug for treating TNBC through miR-374a repression.
Keywords: 3′,4′,5′,4″-tetramethoxychalcone; Bax; apoptosis; miR-374a; triple-negative breast cancer.
Copyright © 2020 Peng, Xiong, Xie, Tang, Huang and Peng.
Figures



References
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

