Progressive Choriocapillaris Impairment in ABCA4 Maculopathy Is Secondary to Retinal Pigment Epithelium Atrophy
- PMID: 32298433
- PMCID: PMC7401499
- DOI: 10.1167/iovs.61.4.13
Progressive Choriocapillaris Impairment in ABCA4 Maculopathy Is Secondary to Retinal Pigment Epithelium Atrophy
Abstract
Purpose: To analyze the progression of choriocapillaris (CC) impairment in recessive Stargardt disease (STGD) and compare it to the progression of retinal pigment epithelium (RPE) atrophy.
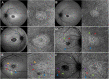
Methods: Fifty-five patients with a clinical diagnosis of STGD and genetic confirmation of pathogenic biallelic variants in ABCA4 were imaged with short-wavelength fundus autofluorescence (SW-AF) and optical coherence tomography angiography (OCTA) at a single clinic visit, whereas a subset of 12 patients were imaged with the same modalities at two different clinic visits.
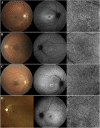
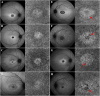
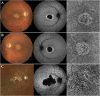
Results: We observed three stages of CC impairment: an area of bright yet intact macular CC (11 patients), regions of vascular rarefaction and incomplete CC atrophy within an area of bright CC (10 patients), and areas of extensive CC atrophy (26 patients). These changes correlated to the degree of RPE atrophy observed in SW-AF imaging. Furthermore, 8 patients presented with early changes on SW-AF, but healthy CC. Quantitative analyses of the atrophic changes revealed that the area of RPE atrophy is larger (9.6 ± 1.7 mm2 vs. 6.9 ± 1.3 mm2, P < 0.001) and that it progresses at a faster rate (1.1 ± 0.1 mm2/year vs. 0.8 ± 0.2 mm2/year, P = 0.004) than the corresponding area of CC atrophy.
Conclusions: CC impairment is progressive and OCTA imaging can be used to demonstrate the stages, which culminate in extensive CC atrophy. Furthermore, CC impairment is secondary to RPE atrophy in STGD. We further advocate the use of SW-AF and OCTA imaging in monitoring the progression of STGD.
Conflict of interest statement
Disclosure:
Figures

References
-
- Allikmets R, Singh N, Sun H, et al.. A photoreceptor cell-specific ATP-binding transporter gene (ABCR) is mutated in recessive Stargardt macular dystrophy. Nat Genet. 1997; 15: 236–246. - PubMed
-
- Jauregui R, Cho GY, Takahashi VKL, et al.. Caring for hereditary childhood retinal blindness. Asia Pac J Ophthalmol (Phila). 2018; 7: 183–191. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

