Depletion of resident muscle stem cells negatively impacts running volume, physical function, and muscle fiber hypertrophy in response to lifelong physical activity
- PMID: 32320286
- PMCID: PMC7311742
- DOI: 10.1152/ajpcell.00090.2020
Depletion of resident muscle stem cells negatively impacts running volume, physical function, and muscle fiber hypertrophy in response to lifelong physical activity
Abstract
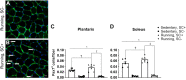
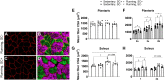
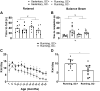
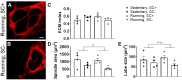
To date, studies that have aimed to investigate the role of satellite cells during adult skeletal muscle adaptation and hypertrophy have utilized a nontranslational stimulus and/or have been performed over a relatively short time frame. Although it has been shown that satellite cell depletion throughout adulthood does not drive skeletal muscle loss in sedentary mice, it remains unknown how satellite cells participate in skeletal muscle adaptation to long-term physical activity. The current study was designed to determine whether reduced satellite cell content throughout adulthood would influence the transcriptome-wide response to physical activity and diminish the adaptive response of skeletal muscle. We administered vehicle or tamoxifen to adult Pax7-diphtheria toxin A (DTA) mice to deplete satellite cells and assigned them to sedentary or wheel-running conditions for 13 mo. Satellite cell depletion throughout adulthood reduced balance and coordination, overall running volume, and the size of muscle proprioceptors (spindle fibers). Furthermore, satellite cell participation was necessary for optimal muscle fiber hypertrophy but not adaptations in fiber type distribution in response to lifelong physical activity. Transcriptome-wide analysis of the plantaris and soleus revealed that satellite cell function is muscle type specific; satellite cell-dependent myonuclear accretion was apparent in oxidative muscles, whereas initiation of G protein-coupled receptor (GPCR) signaling in the glycolytic plantaris may require satellite cells to induce optimal adaptations to long-term physical activity. These findings suggest that satellite cells play a role in preserving physical function during aging and influence muscle adaptation during sustained periods of physical activity.
Keywords: aging; exercise; satellite cells; skeletal muscle; stem cells.
Conflict of interest statement
No conflicts of interest, financial or otherwise, are declared by the authors.
Figures




References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

