The microcircuits of striatum in silico
- PMID: 32321828
- PMCID: PMC7197017
- DOI: 10.1073/pnas.2000671117
The microcircuits of striatum in silico
Abstract
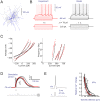
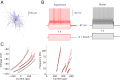
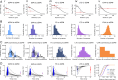
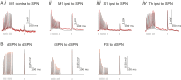
The basal ganglia play an important role in decision making and selection of action primarily based on input from cortex, thalamus, and the dopamine system. Their main input structure, striatum, is central to this process. It consists of two types of projection neurons, together representing 95% of the neurons, and 5% of interneurons, among which are the cholinergic, fast-spiking, and low threshold-spiking subtypes. The membrane properties, soma-dendritic shape, and intrastriatal and extrastriatal synaptic interactions of these neurons are quite well described in the mouse, and therefore they can be simulated in sufficient detail to capture their intrinsic properties, as well as the connectivity. We focus on simulation at the striatal cellular/microcircuit level, in which the molecular/subcellular and systems levels meet. We present a nearly full-scale model of the mouse striatum using available data on synaptic connectivity, cellular morphology, and electrophysiological properties to create a microcircuit mimicking the real network. A striatal volume is populated with reconstructed neuronal morphologies with appropriate cell densities, and then we connect neurons together based on appositions between neurites as possible synapses and constrain them further with available connectivity data. Moreover, we simulate a subset of the striatum involving 10,000 neurons, with input from cortex, thalamus, and the dopamine system, as a proof of principle. Simulation at this biological scale should serve as an invaluable tool to understand the mode of operation of this complex structure. This platform will be updated with new data and expanded to simulate the entire striatum.
Keywords: basal ganglia; compartmental models; computational analysis; modeling; network.
Copyright © 2020 the Author(s). Published by PNAS.
Conflict of interest statement
The authors declare no competing interest.
Figures





Similar articles
-
Excitatory extrinsic afferents to striatal interneurons and interactions with striatal microcircuitry.Eur J Neurosci. 2019 Mar;49(5):593-603. doi: 10.1111/ejn.13881. Epub 2018 Mar 25. Eur J Neurosci. 2019. PMID: 29480942 Free PMC article. Review.
-
Dopamine-modulated dynamic cell assemblies generated by the GABAergic striatal microcircuit.Neural Netw. 2009 Oct;22(8):1174-88. doi: 10.1016/j.neunet.2009.07.018. Epub 2009 Jul 19. Neural Netw. 2009. PMID: 19646846 Review.
-
Activity Dynamics and Signal Representation in a Striatal Network Model with Distance-Dependent Connectivity.eNeuro. 2017 Aug 23;4(4):ENEURO.0348-16.2017. doi: 10.1523/ENEURO.0348-16.2017. eCollection 2017 Jul-Aug. eNeuro. 2017. PMID: 28840190 Free PMC article.
-
Glutamatergic Innervation onto Striatal Neurons Potentiates GABAergic Synaptic Output.J Neurosci. 2019 Jun 5;39(23):4448-4460. doi: 10.1523/JNEUROSCI.2630-18.2019. Epub 2019 Apr 1. J Neurosci. 2019. PMID: 30936241 Free PMC article.
-
Modeling influences of dopamine on synchronization behavior of striatum.Network. 2017;28(1):28-52. doi: 10.1080/0954898X.2017.1378824. Epub 2017 Oct 6. Network. 2017. PMID: 28985088 Review.
Cited by
-
Er81 Transcription Factor Fine-Tunes Striatal Cholinergic Interneuron Activity and Drives Habit Formation.J Neurosci. 2021 May 19;41(20):4392-4409. doi: 10.1523/JNEUROSCI.0967-20.2021. Epub 2021 Apr 13. J Neurosci. 2021. PMID: 33849945 Free PMC article.
-
Multiscale modeling of neuronal dynamics in hippocampus CA1.Front Comput Neurosci. 2024 Aug 6;18:1432593. doi: 10.3389/fncom.2024.1432593. eCollection 2024. Front Comput Neurosci. 2024. PMID: 39165754 Free PMC article.
-
Cortical control of striatal fast-spiking interneuron synchrony.J Physiol. 2022 May;600(9):2189-2202. doi: 10.1113/JP282850. Epub 2022 Apr 11. J Physiol. 2022. PMID: 35332539 Free PMC article.
-
An integrative data-driven model simulating C. elegans brain, body and environment interactions.Nat Comput Sci. 2024 Dec;4(12):978-990. doi: 10.1038/s43588-024-00738-w. Epub 2024 Dec 16. Nat Comput Sci. 2024. PMID: 39681671 Free PMC article.
-
Spiny projection neurons exhibit transcriptional signatures within subregions of the dorsal striatum.Cell Rep. 2023 Nov 28;42(11):113435. doi: 10.1016/j.celrep.2023.113435. Epub 2023 Nov 11. Cell Rep. 2023. PMID: 37952158 Free PMC article.
References
-
- Graybiel A. M., The basal ganglia: Learning new tricks and loving it. Curr. Opin. Neurobiol. 15, 638–644 (2005). - PubMed
-
- Graybiel A. M., Habits, rituals, and the evaluative brain. Annu. Rev. Neurosci. 31, 359–387 (2008). - PubMed
-
- Klaus A., Alves da Silva J., Costa R. M., What, if, and when to move: Basal ganglia circuits and self-paced action initiation. Annu. Rev. Neurosci. 42, 459–483 (2019). - PubMed
-
- Arber S., Costa R. M., Connecting neuronal circuits for movement. Science 360, 1403–1404 (2018). - PubMed
-
- Oorschot D. E., Total number of neurons in the neostriatal, pallidal, subthalamic, and substantia nigral nuclei of the rat basal ganglia: A stereological study using the cavalieri and optical disector methods. J. Comp. Neurol. 366, 580–599 (1996). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

