Non-coding and Loss-of-Function Coding Variants in TET2 are Associated with Multiple Neurodegenerative Diseases
- PMID: 32330418
- PMCID: PMC7212268
- DOI: 10.1016/j.ajhg.2020.03.010
Non-coding and Loss-of-Function Coding Variants in TET2 are Associated with Multiple Neurodegenerative Diseases
Abstract
We conducted genome sequencing to search for rare variation contributing to early-onset Alzheimer's disease (EOAD) and frontotemporal dementia (FTD). Discovery analysis was conducted on 435 cases and 671 controls of European ancestry. Burden testing for rare variation associated with disease was conducted using filters based on variant rarity (less than one in 10,000 or private), computational prediction of deleteriousness (CADD) (10 or 15 thresholds), and molecular function (protein loss-of-function [LoF] only, coding alteration only, or coding plus non-coding variants in experimentally predicted regulatory regions). Replication analysis was conducted on 16,434 independent cases and 15,587 independent controls. Rare variants in TET2 were enriched in the discovery combined EOAD and FTD cohort (p = 4.6 × 10-8, genome-wide corrected p = 0.0026). Most of these variants were canonical LoF or non-coding in predicted regulatory regions. This enrichment replicated across several cohorts of Alzheimer's disease (AD) and FTD (replication only p = 0.0029). The combined analysis odds ratio was 2.3 (95% confidence interval [CI] 1.6-3.4) for AD and FTD. The odds ratio for qualifying non-coding variants considered independently from coding variants was 3.7 (95% CI 1.7-9.4). For LoF variants, the combined odds ratio (for AD, FTD, and amyotrophic lateral sclerosis, which shares clinicopathological overlap with FTD) was 3.1 (95% CI 1.9-5.2). TET2 catalyzes DNA demethylation. Given well-defined changes in DNA methylation that occur during aging, rare variation in TET2 may confer risk for neurodegeneration by altering the homeostasis of key aging-related processes. Additionally, our study emphasizes the relevance of non-coding variation in genetic studies of complex disease.
Keywords: AD; ALS; Alzheimer; FTD; TET2; aging; amyotrophic lateral sclerosis; frontotemporal dementia; genome sequencing; non-coding.
Copyright © 2020 American Society of Human Genetics. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
The authors declare competing interests. G.D.R. industry relationships: Research support from Avid Radiopharmaceuticals, Eli Lilly, GE Healthcare, and Life Molecular Imaging. Consultant/scientific advisory board member for Axon Neurosciences, Eisai, and Merck. Speaking honorarium from GE Healthcare. Associate Editor for JAMA Neurology.
Figures
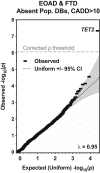

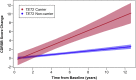
Comment in
-
The Role of Age-Related Clonal Hematopoiesis in Genetic Sequencing Studies.Am J Hum Genet. 2020 Sep 3;107(3):575-576. doi: 10.1016/j.ajhg.2020.07.011. Am J Hum Genet. 2020. PMID: 32888507 Free PMC article. No abstract available.
-
Response to Holstege et al.Am J Hum Genet. 2020 Sep 3;107(3):577-578. doi: 10.1016/j.ajhg.2020.07.012. Am J Hum Genet. 2020. PMID: 32888508 Free PMC article. No abstract available.
References
-
- Lambert M.A., Bickel H., Prince M., Fratiglioni L., Von Strauss E., Frydecka D., Kiejna A., Georges J., Reynish E.L. Estimating the burden of early onset dementia; systematic review of disease prevalence. Eur. J. Neurol. 2014;21:563–569. - PubMed
-
- Loy C.T., Schofield P.R., Turner A.M., Kwok J.B. Genetics of dementia. Lancet. 2014;383:828–840. - PubMed
-
- Goldman J.S., Farmer J.M., Wood E.M., Johnson J.K., Boxer A., Neuhaus J., Lomen-Hoerth C., Wilhelmsen K.C., Lee V.M., Grossman M., Miller B.L. Comparison of family histories in FTLD subtypes and related tauopathies. Neurology. 2005;65:1817–1819. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

