Inducible Kiss1 knockdown in the hypothalamic arcuate nucleus suppressed pulsatile secretion of luteinizing hormone in male mice
- PMID: 32336702
- PMCID: PMC7470898
- DOI: 10.1262/jrd.2019-164
Inducible Kiss1 knockdown in the hypothalamic arcuate nucleus suppressed pulsatile secretion of luteinizing hormone in male mice
Abstract
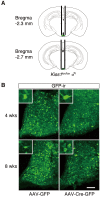
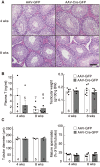
Accumulating evidence suggests that kisspeptin-GPR54 signaling is indispensable for gonadotropin-releasing hormone (GnRH)/gonadotropin secretion and consequent reproductive functions in mammals. Conventional Kiss1 knockout (KO) mice and rats are reported to be infertile. To date, however, no study has investigated the effect of inducible central Kiss1 KO/knockdown on pulsatile gonadotropin release in male mammals. Here we report an in vivo analysis of inducible conditional Kiss1 knockdown male mice. The mice were generated by a bilateral injections of either adeno-associated virus (AAV) vectors driving Cre recombinase (AAV-Cre) or AAV vectors driving GFP (AAV-GFP, control) into the hypothalamic arcuate nucleus (ARC) of Kiss1-floxed male mice, in which exon 3 of the Kiss1 gene were floxed with loxP sites. Four weeks after the AAV-Cre injection, the mice showed a profound decrease in the both number of ARC Kiss1-expressing cells and the luteinizing hormone (LH) pulse frequency. Interestingly, pulsatile LH secretion was apparent 8 weeks after the AAV-Cre injection despite the suppression of ARC Kiss1 expression. The control Kiss1-floxed mice infected with AAV-GFP showed apparent LH pulses and Kiss1 expression in the ARC at both 4 and 8 weeks after the AAV-GFP injection. These results with an inducible conditional Kiss1 knockdown in the ARC of male mice suggest that ARC kisspeptin neurons are responsible for pulsatile LH secretion in male mice, and indicate the possibility of a compensatory mechanism that restores GnRH/LH pulse generation.
Keywords: Adeno-associated virus; Gonadotropin; Gonadotropin-releasing hormone; Kiss1; Metastin.
Conflict of interest statement
The authors have nothing to disclose.
Figures


Similar articles
-
Deficiency of arcuate nucleus kisspeptin results in postpubertal central hypogonadism.Am J Physiol Endocrinol Metab. 2021 Aug 1;321(2):E264-E280. doi: 10.1152/ajpendo.00088.2021. Epub 2021 Jun 28. Am J Physiol Endocrinol Metab. 2021. PMID: 34181485 Free PMC article.
-
GnRH(1-5), a metabolite of gonadotropin-releasing hormone, enhances luteinizing hormone release via activation of kisspeptin neurons in female rats.Endocr J. 2020 Apr 28;67(4):409-418. doi: 10.1507/endocrj.EJ19-0444. Epub 2020 Jan 15. Endocr J. 2020. PMID: 31941848
-
Direct evidence that KNDy neurons maintain gonadotropin pulses and folliculogenesis as the GnRH pulse generator.Proc Natl Acad Sci U S A. 2021 Feb 2;118(5):e2009156118. doi: 10.1073/pnas.2009156118. Proc Natl Acad Sci U S A. 2021. PMID: 33500349 Free PMC article.
-
The role of KNDy neurons in human reproductive health.Endocr J. 2024 Aug 8;71(8):733-743. doi: 10.1507/endocrj.EJ24-0006. Epub 2024 Jun 12. Endocr J. 2024. PMID: 38866494 Review.
-
The roles of kisspeptin in the mechanism underlying reproductive functions in mammals.J Reprod Dev. 2018 Dec 14;64(6):469-476. doi: 10.1262/jrd.2018-110. Epub 2018 Oct 8. J Reprod Dev. 2018. PMID: 30298825 Free PMC article. Review.
Cited by
-
Enkephalin-δ opioid receptor signaling partly mediates suppression of LH release during early lactation in rats.J Reprod Dev. 2023 Aug 11;69(4):192-197. doi: 10.1262/jrd.2023-006. Epub 2023 Jun 16. J Reprod Dev. 2023. PMID: 37331801 Free PMC article.
-
The role of neuroestrogens in the estrogen-induced gonadotropin surge in male monkeys.J Neuroendocrinol. 2024 Oct;36(10):e13413. doi: 10.1111/jne.13413. Epub 2024 May 17. J Neuroendocrinol. 2024. PMID: 38760983
-
KNDy Neurons of the Hypothalamus and Their Role in GnRH Pulse Generation: an Update.Endocrinology. 2023 Dec 23;165(2):bqad194. doi: 10.1210/endocr/bqad194. Endocrinology. 2023. PMID: 38170643 Free PMC article. Review.
-
Central somatostatin-somatostatin receptor 2 signaling mediates lactational suppression of luteinizing hormone release via the inhibition of glutamatergic interneurons during late lactation in rats.J Reprod Dev. 2022 Jun 1;68(3):190-197. doi: 10.1262/jrd.2022-009. Epub 2022 Mar 6. J Reprod Dev. 2022. PMID: 35249910 Free PMC article.
-
Sex difference in developmental changes in visualized Kiss1 neurons in newly generated Kiss1-Cre rats.J Reprod Dev. 2023 Oct 20;69(5):227-238. doi: 10.1262/jrd.2023-019. Epub 2023 Jul 28. J Reprod Dev. 2023. PMID: 37518187 Free PMC article.
References
-
- Ohkura S, Tsukamura H, Maeda K. Effects of various types of hypothalamic deafferentation on luteinizing hormone pulses in ovariectomized rats. J Neuroendocrinol 1991; 3: 503–508. - PubMed
-
- Gore AC, Terasawa E. Neural circuits regulating pulsatile luteinizing hormone release in the female guinea-pig: opioid, adrenergic and serotonergic interactions. J Neuroendocrinol 2001; 13: 239–248. - PubMed
-
- Noguchi M, Yoshioka K, Kaneko H, Iwamura S, Takahashi T, Suzuki C, Arai S, Wada Y, Itoh S. Measurement of porcine luteinizing hormone concentration in blood by time-resolved fluoroimmunoassay. J Vet Med Sci 2007; 69: 1291–1294. - PubMed
-
- Moenter SM, Brand RM, Midgley AR, Karsch FJ. Dynamics of gonadotropin-releasing hormone release during a pulse. Endocrinology 1992; 130: 503–510. - PubMed
-
- Tanaka T, Ozawa T, Hoshino K, Mori Y. Changes in the gonadotropin-releasing hormone pulse generator activity during the estrous cycle in the goat. Neuroendocrinology 1995; 62: 553–561. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

