Cerebellar Purkinje cell activity modulates aggressive behavior
- PMID: 32343225
- PMCID: PMC7202893
- DOI: 10.7554/eLife.53229
Cerebellar Purkinje cell activity modulates aggressive behavior
Abstract
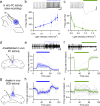
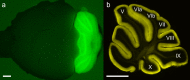
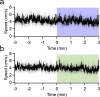
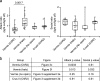
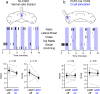
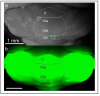
Although the cerebellum is traditionally associated with balance and motor function, it also plays wider roles in affective and cognitive behaviors. Evidence suggests that the cerebellar vermis may regulate aggressive behavior, though the cerebellar circuits and patterns of activity that influence aggression remain unclear. We used optogenetic methods to bidirectionally modulate the activity of spatially-delineated cerebellar Purkinje cells to evaluate the impact on aggression in mice. Increasing Purkinje cell activity in the vermis significantly reduced the frequency of attacks in a resident-intruder assay. Reduced aggression was not a consequence of impaired motor function, because optogenetic stimulation did not alter motor performance. In complementary experiments, optogenetic inhibition of Purkinje cells in the vermis increased the frequency of attacks. These results suggest Purkinje cell activity in the cerebellar vermis regulates aggression, and further support the importance of the cerebellum in driving affective behaviors that could contribute to neurological disorders.
Keywords: aggression; cerebellum; mouse; neuroscience; optogenetics.
© 2020, Jackman et al.
Conflict of interest statement
SJ, CC, HO, ID, BH, AB, KF, IF, WR No competing interests declared
Figures




References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

