Irinotecan, Temozolomide, and Dinutuximab With GM-CSF in Children With Refractory or Relapsed Neuroblastoma: A Report From the Children's Oncology Group
- PMID: 32343642
- PMCID: PMC7325366
- DOI: 10.1200/JCO.20.00203
Irinotecan, Temozolomide, and Dinutuximab With GM-CSF in Children With Refractory or Relapsed Neuroblastoma: A Report From the Children's Oncology Group
Abstract
Purpose: The combination of irinotecan, temozolomide, dintuximab, and granulocyte-macrophage colony-stimulating factor (I/T/DIN/GM-CSF) demonstrated activity in patients with relapsed/refractory neuroblastoma in the randomized Children's Oncology Group ANBL1221 trial. To more accurately assess response rate and toxicity, an expanded cohort was nonrandomly assigned to I/T/DIN/GM-CSF.
Patients and methods: Patients were eligible at first relapse or first designation of refractory disease. Oral T and intravenous (IV) irinotecan were administered on days 1 to 5 of 21-day cycles. DIN was administered IV (days 2-5), and GM-CSF was administered subcutaneously (days 6-12). The primary end point was objective response, analyzed on an intent-to-treat basis per the International Neuroblastoma Response Criteria.
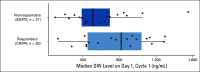
Results: Seventeen eligible patients were randomly assigned to I/T/DIN/GM-CSF (February 2013 to March 2015); 36 additional patients were nonrandomly assigned to I/T/DIN/GM-CSF (August 2016 to May 2017). Objective (complete or partial) responses were observed in nine (52.9%) of 17 randomly assigned patients (95% CI, 29.2% to 76.7%) and 13 (36.1%) of 36 expansion patients (95% CI, 20.4% to 51.8%). Objective responses were seen in 22 (41.5%) of 53 patients overall (95% CI, 28.2% to 54.8%); stable disease was also observed in 22 of 53. One-year progression-free and overall survival for all patients receiving I/T/DIN/GM-CSF were 67.9% ± 6.4% (95% CI, 55.4% to 80.5%) and 84.9% ± 4.9% (95% CI, 75.3% to 94.6%), respectively. Two patients did not receive protocol therapy and were evaluable for response but not toxicity. Common grade ≥ 3 toxicities were fever/infection (18 [35.3%] of 51), neutropenia (17 [33.3%] of 51), pain (15 [29.4%] of 51), and diarrhea (10 [19.6%] of 51). One patient met protocol-defined criteria for unacceptable toxicity (grade 4 hypoxia). Higher DIN trough levels were associated with response.
Conclusion: I/T/DIN/GM-CSF has significant antitumor activity in patients with relapsed/refractory neuroblastoma. Study of chemoimmunotherapy in the frontline setting is indicated, as is further evaluation of predictive biomarkers.
Trial registration: ClinicalTrials.gov NCT01767194.
Figures


References
-
- Ladenstein R, Pötschger U, Pearson ADJ, et al. Busulfan and melphalan versus carboplatin, etoposide, and melphalan as high-dose chemotherapy for high-risk neuroblastoma (HR-NBL1/SIOPEN): An international, randomised, multi-arm, open-label, phase 3 trial. Lancet Oncol. 2017;18:500–514. - PubMed
-
- Coiffier B, Lepage E, Briere J, et al. CHOP chemotherapy plus rituximab compared with CHOP alone in elderly patients with diffuse large-B-cell lymphoma. N Engl J Med. 2002;346:235–242. - PubMed
-
- Forstpointner R, Dreyling M, Repp R, et al. The addition of rituximab to a combination of fludarabine, cyclophosphamide, mitoxantrone (FCM) significantly increases the response rate and prolongs survival as compared with FCM alone in patients with relapsed and refractory follicular and mantle cell lymphomas: Results of a prospective randomized study of the German Low-Grade Lymphoma Study Group. Blood. 2004;104:3064–3071. - PubMed
-
- Haynes NM, van der Most RG, Lake RA, et al. Immunogenic anti-cancer chemotherapy as an emerging concept. Curr Opin Immunol. 2008;20:545–557. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

