Impact of immune enhancement on Covid-19 polyclonal hyperimmune globulin therapy and vaccine development
- PMID: 32344202
- PMCID: PMC7161485
- DOI: 10.1016/j.ebiom.2020.102768
Impact of immune enhancement on Covid-19 polyclonal hyperimmune globulin therapy and vaccine development
Abstract
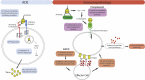
The pandemic spread of a novel coronavirus - SARS coronavirus-2 (SARS-CoV-2) as a cause of acute respiratory illness, named Covid-19, is placing the healthcare systems of many countries under unprecedented stress. Global economies are also spiraling towards a recession in fear of this new life-threatening disease. Vaccines that prevent SARS-CoV-2 infection and therapeutics that reduces the risk of severe Covid-19 are thus urgently needed. A rapid method to derive antiviral treatment for Covid-19 is the use of convalescent plasma derived hyperimmune globulin. However, both hyperimmune globulin and vaccine development face a common hurdle - the risk of antibody-mediated disease enhancement. The goal of this review is to examine the body of evidence supporting the hypothesis of immune enhancement that could be pertinent to Covid-19. We also discuss how this risk could be mitigated so that both hyperimmune globulin and vaccines could be rapidly translated to overcome the current global health crisis.
Keywords: COVID-19; Coronavirus; Polyclonal hyperimmune globulin; SARS-CoV-2; Vaccines.
Copyright © 2020 The Author(s). Published by Elsevier B.V. All rights reserved.
Conflict of interest statement
Declaration of Competing Interest The authors declare no conflict of interest.
Figures
References
-
- WHO . In: Coronavirus disease 2019 (COVID-19) situation report 72. WHO, editor. WHO; Geneva: 2020. editor. WHO.
-
- Guan W.J., Ni Z.Y., Hu Y. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. 2020 Available from: https://www.nejm.org/doi/full/10.1056/NEJMoa2002032. - DOI - PMC - PubMed
-
- Hung I.F.N., To K.K.W., Lee C.K. Hyperimmune IV immunoglobulin treatment: a multicenter double-blind randomized controlled trial for patients with severe 2009 influenza A(H1N1) infection. Chest. 2013;144(2):464–473. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

