Heat Shock Protein 60 in Hepatocellular Carcinoma: Insights and Perspectives
- PMID: 32351972
- PMCID: PMC7174549
- DOI: 10.3389/fmolb.2020.00060
Heat Shock Protein 60 in Hepatocellular Carcinoma: Insights and Perspectives
Abstract
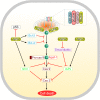
Heat shock protein 60 (HSP60) is a mitochondrial chaperone that is implicated in physiological and pathological processes. For instance, it contributes to protein folding and stability, translocation of mitochondrial proteins, and apoptosis. Variations in the expression levels of HSP60 have been correlated to various diseases and cancers, including hepatocellular carcinoma (HCC). Unlike other HSPs which clearly increase in some cancers, data about HSP60 levels in HCC are controversial and difficult to interpret. In the current review, we summarize and simplify the current knowledge about the role of HSP60 in HCC. In addition, we highlight the possibility of its targeting, using chemical compounds and/or genetic tools for treatment of HCC.
Keywords: cancer therapy; chaperones; chaperonin; heat shock proteins; hepatocellular carcinoma; therapeutic resistance.
Copyright © 2020 Hoter, Rizk and Naim.
Figures

References
-
- Abd El-Salam F. M., El-Sharqawy E. H., El-feky H. M., Mohammed S. A., Edres A. M. (2017). Heat shock protein 60 and chromatin assembly factor-1 mRNA levels in hepatitis C virus-related hepatocellular carcinoma and clinical significance. Int. J. Res. Med. Sci. 5:965 10.18203/2320-6012.ijrms20170644 - DOI
Publication types
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

