The Agnostic Role of Site of Metastasis in Predicting Outcomes in Cancer Patients Treated with Immunotherapy
- PMID: 32353934
- PMCID: PMC7349154
- DOI: 10.3390/vaccines8020203
The Agnostic Role of Site of Metastasis in Predicting Outcomes in Cancer Patients Treated with Immunotherapy
Abstract
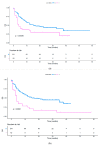
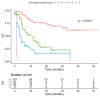
Immune checkpoint inhibitors have revolutionized treatment and outcome of melanoma and many other solid malignancies including non-small cell lung cancer (NSCLC) and renal cell carcinoma (RCC). Unfortunately, only a minority of patients have a long-term benefit, while the remaining demonstrate primary or acquired resistance. Recently, it has been demonstrated that the prevalence of programmed death-ligand 1 (PD-L1) and tumor-infiltrating lymphocytes (TILs) varies based on the anatomical site of metastases. In particular, liver seems to have more immunosuppressive microenvironment while both the presence of lymph nodal disease and lung metastases seem to have the highest prevalence of PD-L1 and TILs. The aim of the present study is to investigate the possible role of site of metastases as a predictive factor for response or resistance to immunotherapy in several types of cancer. In this multicenter retrospective study, we enrolled patients with metastatic NSCLC, melanoma, RCC, urothelial, merkel carcinoma, and colon cancer who received immunotherapy from April 2015 to August 2019. Major clinicopathological parameters were retrieved and correlated with patients' survival outcomes in order to assess their prognostic value and build a useful tool to assist in the decision-making process. A total of 291 patients were included in this study. One hundred eighty-seven (64%) patients were male and 104 (36%) female. The tumor histology was squamous NSCLC in 56 (19%) patients, non-squamous NSCLC in 99 (34%) patients, melanoma in 101 (35%) patients, RCC in 28 (10%) patients, and other tumors in the remaining 7 (2%) patients. The number of metastatic sites was 1 in 103 patients (35%), 2 in 104 patients (36%) and 3 in 84 patients (29%). Out of 183 valuable patients, the entity of response was complete response (CR), partial response (PR), stable disease (SD), and progression disease (PD) in 15, 53, 31, and 79 patients, respectively. Using an univariate analysis (UVA), tumor burden (p = 0.0004), the presence of liver (p = 0.0009), bone (p = 0.0016), brain metastases (p < 0.0001), the other metastatic sites (p = 0.0375), the number of metastatic sites (p = 0.0039), the histology (p = 0.0034), the upfront use of immunotherapy (p = 0.0032), and Eastern Cooperative Oncology Group (ECOG) Perfomance status (PS) ≥ 1 (p < 0.0001) were significantly associated with poor overall survival (OS). Using a multivariate analysis (MVA) the presence of liver (p = 0.0105) and brain (p = 0.0026) metastases, the NSCLC diagnosis (p < 0.0001) and the ECOG PS (p < 0.0001) resulted as significant prognostic factors of survival. Regarding the progression free survival (PFS), using a UVA of the tumor burden (p = 0.0004), bone (p = 0.0098) and brain (p = 0.0038) metastases, the presence of other metastatic sites (p = 0.0063), the number of metastatic sites (p = 0.0007), the histology (p = 0.0007), the use of immunotherapy as first line (p = 0.0031), and the ECOG PS ≥ 1 (p ≤ 0.0001) were associated with a lower PFS rate. Using an MVA, the presence of brain (p = 0.0088) and liver metastases (p = 0.024) and the ECOG PS (p < 0.0001) resulted as predictors of poor PFS. Our study suggests that the site of metastases could have a role as prognostic and predictive factor in patients treated with immunotherapy. Indeed, regardless of the histology, the presence of liver and brain metastases was associated with a shorter PFS and OS, but these results must be confirmed in further studies. In this context, a deep characterization of microenvironment could be crucial to prepare patients through novel strategies with combination or sequential immunotherapy in order to improve treatment response.
Keywords: NSCLC; RCC; agnostic biomarkers; immunotherapy; melanoma; metastatic sites; predictive factors; prognostic factors.
Conflict of interest statement
Paolo Marchetti (PM) has/had a consultant/advisory role for BMS, RocheGenentech, MSD, Novartis, Amgen, Merck Serono, Pierre Fabre, and Incyte. The other authors declare that the research was conducted in absence of any commercial or financial relationship that could be construed as a potential conflict of interest.
Figures







References
-
- Chae Y.K., Arya A., Iams W., Cruz M.R., Chandra S., Choi J., Giles F. Current landscape and future of dual anti-CTLA4 and PD-1/PD-L1 blockade immunotherapy in cancer; lessons learned from clinical trials with melanoma and non-small cell lung cancer (NSCLC) J. Immunother. Cancer. 2018;6:39. doi: 10.1186/s40425-018-0349-3. - DOI - PMC - PubMed
-
- Balar A.V., Galsky M.D., Rosenberg J.E., Powles T., Petrylak D.P., Bellmunt J., Loriot Y., Necchi A., Hoffman-Censits J., Perez-Gracia J.L., et al. Atezolizumab as first-line treatment in cisplatin-ineligible patients with locally advanced and metastatic urothelial carcinoma: A single-arm, multicentre, phase 2 trial. Lancet. 2017;389:67–76. doi: 10.1016/S0140-6736(16)32455-2. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

