Dieckol, a Major Marine Polyphenol, Enhances Non-Rapid Eye Movement Sleep in Mice via the GABAA-Benzodiazepine Receptor
- PMID: 32362829
- PMCID: PMC7181965
- DOI: 10.3389/fphar.2020.00494
Dieckol, a Major Marine Polyphenol, Enhances Non-Rapid Eye Movement Sleep in Mice via the GABAA-Benzodiazepine Receptor
Abstract
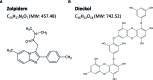
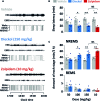
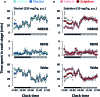
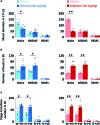
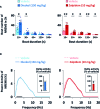
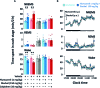
We had previously demonstrated that phlorotannins, which are marine polyphenols, enhance sleep in mice via the GABAA-benzodiazepine (BZD) receptor. Among the constituents of phlorotannin, dieckol is a major marine polyphenol from the brown alga Ecklonia cava. Although phlorotannins are known to exert hypnotic effects, the sleep-enhancing effect of dieckol has not yet been determined. We evaluated the effect of dieckol on sleep-wake state of mice by analyzing electroencephalograms (EEGs) and electromyograms. Flumazenil, a GABAA-BZD antagonist, was used to investigate the molecular mechanism underlying the effects of dieckol on sleep. The polygraphic recordings and corresponding hypnograms revealed that dieckol accelerated the initiation of non-rapid eye movement sleep (NREMS); it shortened sleep latency and increased NREMS duration. According to the change in time-course, dieckol showed sleep-enhancing effects by increasing the amount of NREMS and decreasing wakefulness during the same hours. Additionally, sleep quality was evaluated by analyzing the EEG power density, and dieckol was found to not affect sleep intensity while zolpidem was found to reduce it. Finally, we treated mice with zolpidem or dieckol in combination with flumazenil and found the latter to inhibit the sleep-enhancing effect of dieckol and zolpidem, thereby indicating that dieckol exerts sleep-enhancing effects by activating the GABAA-BZD receptor, similar to zolpidem. These results implied that dieckol can be used as a promising herbal sleep aid with minimal side effects, unlike the existing hypnotics.
Keywords: dieckol; electroencephalogram; hypnotic; marine polyphenols; phlorotannins; sleep.
Copyright © 2020 Yoon, Kim, Seo, Lee, Um, Lee, Jung and Cho.
Figures

References
-
- Barnard E. A., Skolnick P., Olsen R. W., Möhler H., Sieghart W., Biggio G., et al. (1998). International Union of Pharmacology. XV. Subtypes of g-aminobutyric acid A receptors: classification on the basis of subunit structure and receptor function. Pharmacol. Rev. 50, 291–313. - PubMed
-
- Chen C. R., Zhou X. Z., Luo Y. J., Huang Z. L., Urade Y., Qu W. M. (2012). Magnolol, a major bioactive constituent of the bark of Magnolia officinalis, induces sleep via the benzodiazepine site of GABAA receptor in mice. Neuropharmacology 63, 1191–1199. 10.1016/j.neuropharm.2012.06.031 - DOI - PubMed
-
- Cho S., Shimizu M. (2015). “Natural sleep aids and polyphenols as treatments for insomnia,” in Bioactive nutraceuticals and dietary supplements in neurological and brain disease. Eds. Watson R. R., Preedy V. R. (San Diego: Academic Press; ), 141–151.
LinkOut - more resources
Full Text Sources

