The impact of certolizumab pegol treatment on the incidence of anterior uveitis flares in patients with axial spondyloarthritis: 48-week interim results from C-VIEW
- PMID: 32371433
- PMCID: PMC7299504
- DOI: 10.1136/rmdopen-2019-001161
The impact of certolizumab pegol treatment on the incidence of anterior uveitis flares in patients with axial spondyloarthritis: 48-week interim results from C-VIEW
Abstract
Background: Acute anterior uveitis (AAU) is the most common extra-articular manifestation in patients with axial spondyloarthritis (axSpA). C-VIEW investigates the impact of the Fc-free TNF inhibitor certolizumab pegol (CZP) on AAU flares in patients with active axSpA at high risk of recurrent AAU.
Methods: C-VIEW (NCT03020992) is a 96-week ongoing, multicentre, open-label, phase 4 study. Included patients had an axSpA diagnosis, a history of recurrent AAU (≥2 AAU flares, ≥1 flare in the year prior to study entry), HLA-B27 positivity, active disease, and failure of ≥2 non-steroidal anti-inflammatory drugs. Patients received CZP 400 mg at Weeks 0/2/4, then 200 mg every 2 weeks up to 96 weeks. This 48-week pre-planned interim analysis compares AAU flare incidence in the 48 weeks before and after initiation of CZP treatment, using Poisson regression to account for possible within-patient correlations.
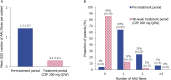
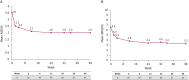
Results: In total, 89 patients were included (male: 63%; radiographic/non-radiographic axSpA: 85%/15%; mean axSpA disease duration: 8.6 years). During 48 weeks' CZP treatment, 13 (15%) patients experienced 15 AAU flares, representing an 87% reduction in AAU incidence rate (146.6 per 100 patient-years (PY) in the 48 weeks pre-baseline to 18.7 per 100 PY during CZP treatment). Poisson regression analysis showed that the incidence rate of AAU per patient reduced from 1.5 to 0.2 (p<0.001). No new safety signals were identified.
Conclusions: There was a significant reduction in the AAU flare rate during 48 weeks of CZP treatment, indicating that CZP is a suitable treatment option for patients with active axSpA and a history of recurrent AAU.
Keywords: TNF inhibitor; axial spondyloarthritis; extra-articular manifestations; uveitis.
© Author(s) (or their employer(s)) 2020. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ.
Conflict of interest statement
Competing interests: Yes, there are competing interests for one or more authors and I have provided a Competing Interests statement in my manuscript.
Figures

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
