Rebound-associated vertebral fractures may occur in sequential time points following denosumab discontinuation: need for prompt treatment re-initiation
- PMID: 32373677
- PMCID: PMC7191536
- DOI: 10.1016/j.bonr.2020.100267
Rebound-associated vertebral fractures may occur in sequential time points following denosumab discontinuation: need for prompt treatment re-initiation
Abstract
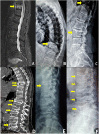
Denosumab discontinuation has been associated with increased risk of rebound-associated multiple vertebral fractures. We report the cases of three patients, two females and one male, who had manifested rebound-associated vertebral fractures after denosumab discontinuation and sustained new vertebral fractures a few months later. Two of the patients had been previously treated with bisphosphonates. Patients discontinued denosumab after 2 to 8 years of treatment. One of the female patients was receiving prednisolone 7.5 mg daily for an unspecified connective tissue disorder and the male patient methylprednisolone 8 mg daily for dermatomyositis. We hypothesize that rebound-associated multiple vertebral fractures after denosumab discontinuation may occur, at least in some cases, sequentially instead of simultaneously. Our cases further underpin the need for prompt initiation of potent antiresorptives in patients who sustained rebound-associated vertebral fractures, in order to prevent not only bone loss but also a second round of fractures.
Keywords: Denosumab; Discontinuation; Fracture; Osteoporosis; Rebound.
© 2020 Published by Elsevier Inc.
Conflict of interest statement
A.D. Anastasilakis reports lecture fees from Amgen, Bianex, Eli-Lilly and ITF; G. Evangelatos has nothing to declare; Makras reports honoraria for lectures and research grants from Amgen; lecture fees from Glaxo, Lilly, Pfizer, Leo, Genesis, Elpen, and Vianex. A. Iliopoulos reports lecture fees from Amgen, MSD, and Novartis.
Figures
References
-
- Anastasilakis A.D., Yavropoulou M.P., Makras P., Sakellariou G.T., Papadopoulou F., Gerou S., Papapoulos S.E. Increased osteoclastogenesis in patients with vertebral fractures following discontinuation of denosumab treatment. Eur. J. Endocrinol. 2017;176:677–683. - PubMed
-
- Anastasilakis A.D., Polyzos S.A., Makras P., Aubry-Rozier B., Kaouri S., Lamy O. Clinical features of 24 patients with rebound-associated vertebral fractures after denosumab discontinuation: systematic review and additional cases. J. Bone Miner. Res. 2017;32:1291–1296. - PubMed
-
- Anastasilakis A.D., Polyzos S.A., Makras P. THERAPY OF ENDOCRINE DISEASE: Denosumab vs bisphosphonates for the treatment of postmenopausal osteoporosis. Eur. J. Endocrinol. 2018;179:R31–R45. - PubMed
-
- Anastasilakis A.D., Papapoulos S.E., Polyzos S.A., Appelman-Dijkstra N.M., Makras P. Zoledronate for the prevention of Bone loss in women discontinuing denosumab treatment. A prospective 2-year clinical trial. J. Bone Miner. Res. 2019;34:2220–2228. - PubMed
-
- Bekker P.J., Holloway D.L., Rasmussen A.S., Murphy R., Martin S.W., Leese P.T., Holmes G.B., Dunstan C.R., DePaoli A.M. A single-dose placebo-controlled study of AMG 162, a fully human monoclonal antibody to RANKL, in postmenopausal women. J. Bone Miner. Res. 2004;19:1059–1066. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Medical

