Impaired Autophagic Activity Contributes to the Pathogenesis of Bronchopulmonary Dysplasia. Evidence from Murine and Baboon Models
- PMID: 32374619
- PMCID: PMC7462342
- DOI: 10.1165/rcmb.2019-0445OC
Impaired Autophagic Activity Contributes to the Pathogenesis of Bronchopulmonary Dysplasia. Evidence from Murine and Baboon Models
Abstract
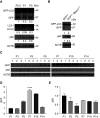
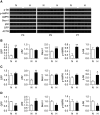
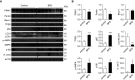
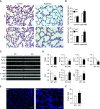
Bronchopulmonary dysplasia (BPD) is a common and serious complication associated with preterm birth. The pathogenesis of BPD is incompletely understood, and there is an unmet clinical need for effective treatments. The role of autophagy as a potential cytoprotective mechanism in BPD remains to be fully elucidated. In the present study, we investigated the role and regulation of autophagy in experimental models of BPD. Regulation and cellular distribution of autophagic activity during postnatal lung development and in neonatal hyperoxia-induced lung injury (nHILI) were assessed in the autophagy reporter transgenic GFP-LC3 (GFP-microtubule-associated protein 1A/1B-light chain 3) mouse model. Autophagic activity and its regulation were also examined in a baboon model of BPD. The role of autophagy in nHILI was determined by assessing lung morphometry, injury, and inflammation in autophagy-deficient Beclin 1 heterozygous knockout mice (Becn1+/-). Autophagic activity was induced during alveolarization in control murine lungs and localized primarily to alveolar type II cells and macrophages. Hyperoxia exposure of neonatal murine lungs and BPD in baboon lungs resulted in impaired autophagic activity in association with insufficient AMPK (5'-AMP-activated protein kinase) and increased mTORC1 (mTOR complex 1) activation. Becn1+/- lungs displayed impaired alveolarization, increased alveolar septal thickness, greater neutrophil accumulation, and increased IL-1β concentrations when exposed to nHILI. Becn1+/- alveolar macrophages isolated from nHILI-exposed mice displayed increased expression of proinflammatory genes. In conclusion, basal autophagy is induced during alveolarization and disrupted during progression of nHILI in mice and BPD in baboons. Becn1+/- mice are more susceptible to nHILI, suggesting that preservation of autophagic activity may be an effective protective strategy in BPD.
Keywords: GFP-LC3; autophagy; beclin; bronchopulmonary dysplasia; macrophage.
Figures

Similar articles
-
AMPK-driven Macrophage Responses Are Autophagy Dependent in Experimental Bronchopulmonary Dysplasia.Am J Respir Cell Mol Biol. 2023 Mar;68(3):279-287. doi: 10.1165/rcmb.2022-0282OC. Am J Respir Cell Mol Biol. 2023. PMID: 36306501 Free PMC article.
-
Inhibition of Regulatory-Associated Protein of Mechanistic Target of Rapamycin Prevents Hyperoxia-Induced Lung Injury by Enhancing Autophagy and Reducing Apoptosis in Neonatal Mice.Am J Respir Cell Mol Biol. 2016 Nov;55(5):722-735. doi: 10.1165/rcmb.2015-0349OC. Am J Respir Cell Mol Biol. 2016. PMID: 27374190 Free PMC article.
-
Hyperoxia reduces STX17 expression and inhibits the autophagic flux in alveolar type II epithelial cells in newborn rats.Int J Mol Med. 2020 Aug;46(2):773-781. doi: 10.3892/ijmm.2020.4617. Epub 2020 May 27. Int J Mol Med. 2020. PMID: 32467992 Free PMC article.
-
The Role of Sphingolipid Signaling in Oxidative Lung Injury and Pathogenesis of Bronchopulmonary Dysplasia.Int J Mol Sci. 2022 Jan 23;23(3):1254. doi: 10.3390/ijms23031254. Int J Mol Sci. 2022. PMID: 35163176 Free PMC article. Review.
-
Hyperoxia-induced bronchopulmonary dysplasia: better models for better therapies.Dis Model Mech. 2021 Feb 23;14(2):dmm047753. doi: 10.1242/dmm.047753. Dis Model Mech. 2021. PMID: 33729989 Free PMC article. Review.
Cited by
-
AMPK-driven Macrophage Responses Are Autophagy Dependent in Experimental Bronchopulmonary Dysplasia.Am J Respir Cell Mol Biol. 2023 Mar;68(3):279-287. doi: 10.1165/rcmb.2022-0282OC. Am J Respir Cell Mol Biol. 2023. PMID: 36306501 Free PMC article.
-
CircRNA, lncRNA, and mRNA profiles of umbilical cord blood exosomes from preterm newborns showing bronchopulmonary dysplasia.Eur J Pediatr. 2022 Sep;181(9):3345-3365. doi: 10.1007/s00431-022-04544-2. Epub 2022 Jul 5. Eur J Pediatr. 2022. PMID: 35790551 Free PMC article.
-
Development and validation of the prediction model based on autophagy-associated genes in bronchopulmonary dysplasia.Ann Med. 2024 Dec;56(1):2433677. doi: 10.1080/07853890.2024.2433677. Epub 2024 Nov 29. Ann Med. 2024. PMID: 39611552 Free PMC article.
-
Comparative analysis of ACE2 protein expression in rodent, non-human primate, and human respiratory tract at baseline and after injury: A conundrum for COVID-19 pathogenesis.PLoS One. 2021 Feb 24;16(2):e0247510. doi: 10.1371/journal.pone.0247510. eCollection 2021. PLoS One. 2021. PMID: 33626084 Free PMC article.
-
Molecular mechanisms of cell death in bronchopulmonary dysplasia.Apoptosis. 2023 Feb;28(1-2):39-54. doi: 10.1007/s10495-022-01791-4. Epub 2022 Nov 11. Apoptosis. 2023. PMID: 36369365 Review.
References
-
- Abman SH, Bancalari E, Jobe A. The evolution of bronchopulmonary dysplasia after 50 years. Am J Respir Crit Care Med. 2017;195:421–424. - PubMed
-
- Stoll BJ, Hansen NI, Bell EF, Walsh MC, Carlo WA, Shankaran S, et al. Eunice Kennedy Shriver National Institute of Child Health and Human Development Neonatal Research Network. Trends in care practices, morbidity, and mortality of extremely preterm neonates, 1993–2012. JAMA. 2015;314:1039–1051. - PMC - PubMed
-
- Abman SH. The dysmorphic pulmonary circulation in bronchopulmonary dysplasia: a growing story. Am J Respir Crit Care Med. 2008;178:114–115. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

