Dysregulation of FOXO1 (Forkhead Box O1 Protein) Drives Calcification in Arterial Calcification due to Deficiency of CD73 and Is Present in Peripheral Artery Disease
- PMID: 32375544
- PMCID: PMC7310306
- DOI: 10.1161/ATVBAHA.119.313765
Dysregulation of FOXO1 (Forkhead Box O1 Protein) Drives Calcification in Arterial Calcification due to Deficiency of CD73 and Is Present in Peripheral Artery Disease
Abstract
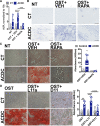
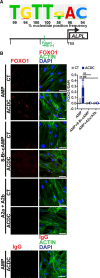
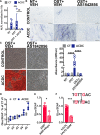
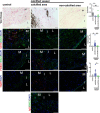
Objective: The recessive disease arterial calcification due to deficiency of CD73 (ACDC) presents with extensive nonatherosclerotic medial layer calcification in lower extremity arteries. Lack of CD73 induces a concomitant increase in TNAP (tissue nonspecific alkaline phosphatase; ALPL), a key enzyme in ectopic mineralization. Our aim was to investigate how loss of CD73 activity leads to increased ALPL expression and calcification in CD73-deficient patients and assess whether this mechanism may apply to peripheral artery disease calcification. Approach and Results: We previously developed a patient-specific disease model using ACDC primary dermal fibroblasts that recapitulates the calcification phenotype in vitro. We found that lack of CD73-mediated adenosine signaling reduced cAMP production and resulted in increased activation of AKT. The AKT/mTOR (mammalian target of rapamycin) axis blocks autophagy and inducing autophagy prevented calcification; however, we did not observe autophagy defects in ACDC cells. In silico analysis identified a putative FOXO1 (forkhead box O1 protein) binding site in the human ALPL promoter. Exogenous AMP induced FOXO1 nuclear localization in ACDC but not in control cells, and this was prevented with a cAMP analogue or activation of A2a/2b adenosine receptors. Inhibiting FOXO1 reduced ALPL expression and TNAP activity and prevented calcification. Mutating the FOXO1 binding site reduced ALPL promoter activation. Importantly, we provide evidence that non-ACDC calcified femoropopliteal arteries exhibit decreased CD73 and increased FOXO1 levels compared with control arteries.
Conclusions: These data show that lack of CD73-mediated cAMP signaling promotes expression of the human ALPL gene via a FOXO1-dependent mechanism. Decreased CD73 and increased FOXO1 was also observed in more common peripheral artery disease calcification.
Keywords: alkaline phosphatase; arteries; calcification; lower extremity.
Figures

Comment in
-
Reining in Peripheral Arterial Calcification.Arterioscler Thromb Vasc Biol. 2020 Jul;40(7):1614-1616. doi: 10.1161/ATVBAHA.120.314665. Epub 2020 Jun 24. Arterioscler Thromb Vasc Biol. 2020. PMID: 32579476 No abstract available.
References
-
- Detrano R, Guerci AD, Carr JJ, Bild DE, Burke G, Folsom AR, Liu K, Shea S, Szklo M, Bluemke DA, et al. Coronary calcium as a predictor of coronary events in four racial or ethnic groups. N Engl J Med. 2008;358:1336–1345. doi: 10.1056/NEJMoa072100. - PubMed
-
- Rutsch F, Ruf N, Vaingankar S, Toliat MR, Suk A, Höhne W, Schauer G, Lehmann M, Roscioli T, Schnabel D, et al. Mutations in ENPP1 are associated with ‘idiopathic’ infantile arterial calcification. Nat Genet. 2003;34:379–381. doi: 10.1038/ng1221. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

