Hippocampal Shape Is Associated with Memory Deficits in Temporal Lobe Epilepsy
- PMID: 32379905
- PMCID: PMC8432153
- DOI: 10.1002/ana.25762
Hippocampal Shape Is Associated with Memory Deficits in Temporal Lobe Epilepsy
Abstract
Objective: Cognitive problems, especially disturbances in episodic memory, and hippocampal sclerosis are common in temporal lobe epilepsy (TLE), but little is known about the relationship of hippocampal morphology with memory. We aimed to relate hippocampal surface-shape patterns to verbal and visual learning.
Methods: We analyzed hippocampal surface shapes on high-resolution magnetic resonance images and the Adult Memory and Information Processing Battery in 145 unilateral refractory TLE patients undergoing epilepsy surgery, a validation set of 55 unilateral refractory TLE patients, and 39 age- and sex-matched healthy volunteers.
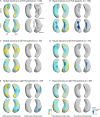
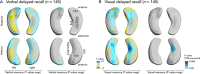
Results: Both left TLE (LTLE) and right TLE (RTLE) patients had lower verbal (LTLE 44 ± 11; RTLE 45 ± 10) and visual learning (LTLE 34 ± 8, RTLE 30 ± 8) scores than healthy controls (verbal 58 ± 8, visual 39 ± 6; p < 0.001). Verbal learning was more impaired the greater the atrophy of the left superolateral hippocampal head. In contrast, visual memory was worse with greater bilateral inferomedial hippocampal atrophy. Postsurgical verbal memory decline was more common in LTLE than in RTLE (reliable change index in LTLE 27% vs RTLE 7%, p = 0.006), whereas there were no differences in postsurgical visual memory decline between those groups. Preoperative atrophy of the left hippocampal tail predicted postsurgical verbal memory decline.
Interpretation: Memory deficits in TLE are associated with specific morphological alterations of the hippocampus, which could help stratify TLE patients into those at high versus low risk of presurgical or postsurgical memory deficits. This knowledge could improve planning and prognosis of selective epilepsy surgery and neuropsychological counseling in TLE. ANN NEUROL 2020 ANN NEUROL 2020;88:170-182.
© 2020 The Authors. Annals of Neurology published by Wiley Periodicals, Inc. on behalf of American Neurological Association.
Conflict of interest statement
Nothing to report.
Figures


References
-
- Helmstaedter C, Kockelmann E. Cognitive outcomes in patients with chronic temporal lobe epilepsy. Epilepsia 2006;47:96–98. - PubMed
-
- Frisk V, Milner B. The role of the left hippocampal region in the acquisition and retention of story content. Neuropsychologia 1990;28:349–359. - PubMed
-
- SML, Milner B. The role of the right hippocampus in the recall of spatial location. Neuropsychologia 1981;19:781–793. - PubMed
-
- Saling MM. Verbal memory in mesial temporal lobe epilepsy: beyond material specificity. Brain 2009;132:570–582. - PubMed
-
- Baxendale S, Thompson P. Defining meaningful postoperative change in epilepsy surgery patients: measuring the unmeasurable? Epilepsy Behav 2005;6:207–211. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

