Chromatin Regulator CHD1 Remodels the Immunosuppressive Tumor Microenvironment in PTEN-Deficient Prostate Cancer
- PMID: 32385075
- PMCID: PMC7483306
- DOI: 10.1158/2159-8290.CD-19-1352
Chromatin Regulator CHD1 Remodels the Immunosuppressive Tumor Microenvironment in PTEN-Deficient Prostate Cancer
Abstract
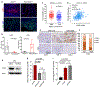
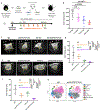
Genetic inactivation of PTEN is common in prostate cancer and correlates with poorer prognosis. We previously identified CHD1 as an essential gene in PTEN-deficient cancer cells. Here, we sought definitive in vivo genetic evidence for, and mechanistic understanding of, the essential role of CHD1 in PTEN-deficient prostate cancer. In Pten and Pten/Smad4 genetically engineered mouse models, prostate-specific deletion of Chd1 resulted in markedly delayed tumor progression and prolonged survival. Chd1 deletion was associated with profound tumor microenvironment (TME) remodeling characterized by reduced myeloid-derived suppressor cells (MDSC) and increased CD8+ T cells. Further analysis identified IL6 as a key transcriptional target of CHD1, which plays a major role in recruitment of immunosuppressive MDSCs. Given the prominent role of MDSCs in suppressing responsiveness to immune checkpoint inhibitors (ICI), our genetic and tumor biological findings support combined testing of anti-IL6 and ICI therapies, specifically in PTEN-deficient prostate cancer. SIGNIFICANCE: We demonstrate a critical role of CHD1 in MDSC recruitment and discover CHD1/IL6 as a major regulator of the immunosuppressive TME of PTEN-deficient prostate cancer. Pharmacologic inhibition of IL6 in combination with immune checkpoint blockade elicits robust antitumor responses in prostate cancer.This article is highlighted in the In This Issue feature, p. 1241.
©2020 American Association for Cancer Research.
Conflict of interest statement
Figures



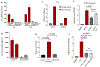

References
-
- Pourmand G, Ziaee AA, Abedi AR, Mehrsai A, Alavi HA, Ahmadi A, et al. Role of PTEN gene in progression of prostate cancer. Urology journal 2007;4(2):95–100. - PubMed
-
- Wang S, Gao J, Lei Q, Rozengurt N, Pritchard C, Jiao J, et al. Prostate-specific deletion of the murine Pten tumor suppressor gene leads to metastatic prostate cancer. Cancer Cell 2003;4(3):209–21 doi S1535610803002150 [pii]. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

