The moonlighting protein c-Fos activates lipid synthesis in neurons, an activity that is critical for cellular differentiation and cortical development
- PMID: 32385110
- PMCID: PMC7324492
- DOI: 10.1074/jbc.RA119.010129
The moonlighting protein c-Fos activates lipid synthesis in neurons, an activity that is critical for cellular differentiation and cortical development
Abstract
Differentiation of neuronal cells is crucial for the development and function of the nervous system. This process involves high rates of membrane expansion, during which the synthesis of membrane lipids must be tightly regulated. In this work, using a variety of molecular and biochemical assays and approaches, including immunofluorescence microscopy and FRET analyses, we demonstrate that the proto-oncogene c-Fos (c-Fos) activates cytoplasmic lipid synthesis in the central nervous system and thereby supports neuronal differentiation. Specifically, in hippocampal primary cultures, blocking c-Fos expression or its activity impairs neuronal differentiation. When examining its subcellular localization, we found that c-Fos co-localizes with endoplasmic reticulum markers and strongly interacts with lipid-synthesizing enzymes, whose activities were markedly increased in vitro in the presence of recombinant c-Fos. Of note, the expression of c-Fos dominant-negative variants capable of blocking its lipid synthesis-activating activity impaired neuronal differentiation. Moreover, using an in utero electroporation model, we observed that neurons with blocked c-Fos expression or lacking its AP-1-independent activity fail to initiate cortical development. These results highlight the importance of c-Fos-mediated activation of lipid synthesis for proper nervous system development.
Keywords: AP-1; AP1 transcription factor (AP-1); c-Fos; central nervous system; central nervous system (CNS); hippocampus; lipid synthesis; neurodevelopment; neurodifferentiation; oncogene; oncogene.; phospholipid metabolism.
© 2020 Rodríguez-Berdini et al.
Conflict of interest statement
Conflict of interest—The authors declare that they have no conflicts of interest with the contents of this article.
Figures
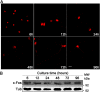


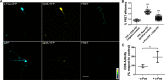


Similar articles
-
c-Fos activated phospholipid synthesis is required for neurite elongation in differentiating PC12 cells.Mol Biol Cell. 2004 Apr;15(4):1881-94. doi: 10.1091/mbc.e03-09-0705. Epub 2004 Feb 6. Mol Biol Cell. 2004. PMID: 14767061 Free PMC article.
-
Mitogen activated protein kinase-dependent activation of c-Jun and c-Fos is required for neuronal differentiation but not for growth and stress response in PC12 cells.J Cell Physiol. 2007 Feb;210(2):538-48. doi: 10.1002/jcp.20907. J Cell Physiol. 2007. PMID: 17111371
-
c-Fos-activated synthesis of nuclear phosphatidylinositol 4,5-bisphosphate [PtdIns(4,5)P₂] promotes global transcriptional changes.Biochem J. 2014 Aug 1;461(3):521-30. doi: 10.1042/BJ20131376. Biochem J. 2014. PMID: 24819416
-
c-Fos: an AP-1 transcription factor with an additional cytoplasmic, non-genomic lipid synthesis activation capacity.Biochim Biophys Acta. 2014 Sep;1841(9):1241-6. doi: 10.1016/j.bbalip.2014.05.007. Epub 2014 Jun 2. Biochim Biophys Acta. 2014. PMID: 24886961 Review.
-
Lipid Metabolism in Neurons: A Brief Story of a Novel c-Fos-Dependent Mechanism for the Regulation of Their Synthesis.Front Cell Neurosci. 2019 May 7;13:198. doi: 10.3389/fncel.2019.00198. eCollection 2019. Front Cell Neurosci. 2019. PMID: 31133814 Free PMC article. Review.
Cited by
-
Axonal Organelles as Molecular Platforms for Axon Growth and Regeneration after Injury.Int J Mol Sci. 2021 Feb 11;22(4):1798. doi: 10.3390/ijms22041798. Int J Mol Sci. 2021. PMID: 33670312 Free PMC article. Review.
-
Combinative Protein Expression of Immediate Early Genes c-Fos, Arc, and Npas4 Along Aversive and Appetitive Experience-Related Neural Networks.Hippocampus. 2025 Sep;35(5):e70030. doi: 10.1002/hipo.70030. Hippocampus. 2025. PMID: 40778693 Free PMC article.
-
Hypo-glycosylated hFSH drives ovarian follicular development more efficiently than fully-glycosylated hFSH: enhanced transcription and PI3K and MAPK signaling.Hum Reprod. 2021 Jun 18;36(7):1891-1906. doi: 10.1093/humrep/deab135. Hum Reprod. 2021. PMID: 34059912 Free PMC article.
-
Phenotype screens of murine pancreatic cancer identify a Tgf-α-Ccl2-paxillin axis driving human-like neural invasion.J Clin Invest. 2023 Nov 1;133(21):e166333. doi: 10.1172/JCI166333. J Clin Invest. 2023. PMID: 37607005 Free PMC article.
-
Endothelial Cell Dysfunction and Hypoxia as Potential Mediators of Pain in Fabry Disease: A Human-Murine Translational Approach.Int J Mol Sci. 2023 Oct 21;24(20):15422. doi: 10.3390/ijms242015422. Int J Mol Sci. 2023. PMID: 37895103 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

