Combining Social Media and FDA Adverse Event Reporting System to Detect Adverse Drug Reactions
- PMID: 32385840
- PMCID: PMC7434724
- DOI: 10.1007/s40264-020-00943-2
Combining Social Media and FDA Adverse Event Reporting System to Detect Adverse Drug Reactions
Abstract
Introduction: Adverse drug reactions (ADRs) are unintended reactions caused by a drug or combination of drugs taken by a patient. The current safety surveillance system relies on spontaneous reporting systems (SRSs) and more recently on observational health data; however, ADR detection may be delayed and lack geographic diversity. The broad scope of social media conversations, such as those on Twitter, can include health-related topics. Consequently, these data could be used to detect potentially novel ADRs with less latency. Although research regarding ADR detection using social media has made progress, findings are based on single information sources, and no study has yet integrated drug safety evidence from both an SRS and Twitter.
Objective: The aim of this study was to combine signals from an SRS and Twitter to facilitate the detection of safety signals and compare the performance of the combined system with signals generated by individual data sources.
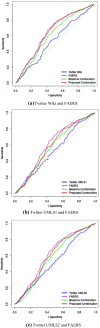
Methods: We extracted potential drug-ADR posts from Twitter, used Monte Carlo expectation maximization to generate drug safety signals from both the US FDA Adverse Event Reporting System and posts from Twitter, and then integrated these signals using a Bayesian hierarchical model. The results from the integrated system and two individual sources were evaluated using a reference standard derived from drug labels. Area under the receiver operating characteristics curve (AUC) was computed to measure performance.
Results: We observed a significant improvement in the AUC of the combined system when comparing it with Twitter alone, and no improvement when comparing with the SRS alone. The AUCs ranged from 0.587 to 0.637 for the combined SRS and Twitter, from 0.525 to 0.534 for Twitter alone, and from 0.612 to 0.642 for the SRS alone. The results varied because different preprocessing procedures were applied to Twitter.
Conclusion: The accuracy of signal detection using social media can be improved by combining signals with those from SRSs. However, the combined system cannot achieve better AUC performance than data from FAERS alone, which may indicate that Twitter data are not ready to be integrated into a purely data-driven combination system.
Conflict of interest statement
Ying Li and Antonio Jimeno Yepes are employed by IBM Research. Cao Xiao is employed by IQVIA. All authors have no conflicts of interest that are directly relevant to the content of this study.
Figures

References
-
- Hauben M, Madigan D, Gerrits CM, Walsh L, Van Puijenbroek EP. The role of data mining in pharmacovigilance. 2005. - PubMed
-
- Powell GE, Seifert HA, Reblin T, Burstein PJ, Blowers J, Menius JA, et al. Social media listening for routine post-marketing safety surveillance. Drug Saf. 2016;39(5):443–454. - PubMed
-
- van der Heijden PG, van Puijenbroek EP, van Buuren S, van der Hofstede JW. On the assessment of adverse drug reactions from spontaneous reporting systems: the influence of under-reporting on odds ratios. Stat Med. 2002;21(14):2027–2044. - PubMed
-
- Poluzzi E, Raschi E, Piccinni C, De Ponti F. Data mining techniques in pharmacovigilance: analysis of the publicly accessible FDA adverse event reporting system (AERS). Data mining applications in engineering and medicine. IntechOpen; 2012.
Publication types
MeSH terms
Associated data
LinkOut - more resources
Full Text Sources
Research Materials

