Synthesis and Thermal Properties of Resorcinol-Furfural Thermosetting Resin
- PMID: 32391489
- PMCID: PMC7203984
- DOI: 10.1021/acsomega.0c00365
Synthesis and Thermal Properties of Resorcinol-Furfural Thermosetting Resin
Abstract
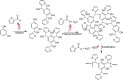
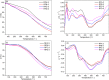
A mild and effective synthesis of resorcinol-furfural (RF) thermosetting resin was proposed with ethanol as a distinctive solvent, which, as a usually neglected factor, was shown to not only help form a homogeneous reaction system but also observably reduce the energy barriers between the early intermediates and transition states in addition reactions by explicit solvent effects, drawn from theoretical calculation conclusions. Besides, the para-additions on aromatic rings were more dominant than ortho-additions with the same reactants, which affected the final link types of monomers verified by Fourier transform infrared spectroscopy and two-dimensional nuclear magnetic resonance tests. The prepared resin can be assigned to a relatively fast gel speed and a high residual mass (65.25%) after pyrolysis in a N2 atmosphere by adjusting the molar ratios of F to R, and the curing of that was a complex reaction, with a curing temperature around 149 °C and an activation energy of about 49.11 kJ mol-1 obtained by the Kissinger method.
Copyright © 2020 American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures




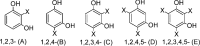


References
-
- Mougel C.; Garnier T.; Cassagnau P.; Sintes-Zydowicz N. Phenolic foams: A review of mechanical properties, fire resistance and new trends in phenol substitution. Polymer 2019, 164, 86–117. 10.1016/j.polymer.2018.12.050. - DOI
-
- Biedermann M.; Grob K. Phenolic resins for can coatings: I. Phenol-based resole analysed by GC–MS, GC×GC, NPLC–GC and SEC. LWT–Food Sci. Technol. 2006, 39, 633–646. 10.1016/j.lwt.2005.04.008. - DOI
-
- Sturiale A.; Vázquez A.; Cisilino A.; Manfredi L. B. Enhancement of the adhesive joint strength of the epoxy–amine system via the addition of a resole-type phenolic resin. Int. J. Adhes. Adhes. 2007, 27, 156–164. 10.1016/j.ijadhadh.2006.01.007. - DOI
-
- Schwan M.; Ratke L. Flexibilisation of resorcinol–formaldehyde aerogels. J. Mater. Chem. A 2013, 1, 13462–13468. 10.1039/c3ta13172f. - DOI
-
- Liu J.; Zhang L.; Yang C.; Tao S. Preparation of multifunctional porous carbon electrodes through direct laser writing on a phenolic resin film. J. Mater. Chem. A 2019, 7, 21168–21175. 10.1039/C9TA07395G. - DOI
LinkOut - more resources
Full Text Sources

